06.02.15
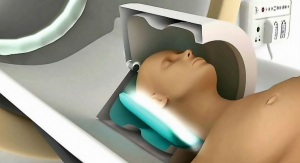
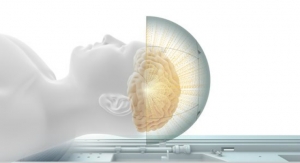
Medtronic plc has secured European regulatory approval for systems within its Activa portfolio of deep-brain stimulation (DBS) neurostimulators for MR conditional full-body magnetic resonance imaging (MRI). The expanded approval for full-body MRI scans applies to all patients receiving a new system and to an estimated 13,000 people in Europe who already have received Medtronic DBS therapy.
Medtronic DBS systems previously have been approved for MRI scans of the head only, under limited conditions. Medtronic DBS systems are not approved in the United States for full-body scans.
MRI scans have become a diagnostic standard of care, allowing physicians to detect a wide range of health conditions by viewing highly detailed images of tumors, internal organs, blood vessels, muscles, joints and other areas of the body by using strong magnetic fields and radio frequency pulses to create images of structures inside the body. Worldwide, it is estimated that approximately 60 million MRI procedures are performed each year, according to figures cited by Medtronic.
When programmed to appropriate settings, MR-conditional Medtronic DBS systems allow patients to continue to receive therapy during MRI scans. Previously, patients receiving an MRI scan had their DBS systems turned off before the scan.
“MRI is commonly the method of choice to image the body to diagnose disease or monitor existing conditions, but MRI use has often been limited in patients receiving DBS therapy,” said John Thornton, medical physicist at the National Hospital for Neurology and Neurosurgery in London, the United Kingdom. “Patients receiving DBS therapy can now receive more advantages of MRI technology.”
“Most patients who we consider for a DBS implant have other conditions which may require MRI,” said Ludvic Zrinzo, neurosurgeon at the National Hospital for Neurology and Neurosurgery in London. “The MR Conditional Activa systems mean patients can receive DBS care, and still may have the option of MRI when needed to manage other conditions.”
To receive approval for MR conditional DBS systems, Medtronic developed proprietary test and measurement systems, in conjunction with advanced electromagnetic modeling tools. Existing Activa DBS systems were tested and evaluated across millions of simulated patient scans spanning more than 38,000 unique implant conditions to demonstrate patient safety.
DBS therapy uses a surgically implanted medical device, similar to a pacemaker, to deliver mild electrical pulses to precisely targeted areas of the brain. The stimulation can be programmed and adjusted noninvasively by a trained clinician to maximize symptom control and minimize side effects. More than 125,000 patients worldwide have received Medtronic DBS Therapy. The therapy currently is approved in many locations around the world, including Europe and the United States, for the treatment of the disabling symptoms of essential tremor, advanced Parkinson’s disease and chronic intractable primary dystonia, for which approval in the United States is under a humanitarian device exemption (HDE). In Europe, Canada and Australia, DBS therapy is approved for the treatment of refractory epilepsy. DBS therapy also is approved for the treatment of severe, treatment-resistant obsessive-compulsive disorder in the European Union and Australia, and in the United States under an HDE.
Medtronic plc is headquartered in Dublin, Ireland.
Medtronic DBS systems previously have been approved for MRI scans of the head only, under limited conditions. Medtronic DBS systems are not approved in the United States for full-body scans.
MRI scans have become a diagnostic standard of care, allowing physicians to detect a wide range of health conditions by viewing highly detailed images of tumors, internal organs, blood vessels, muscles, joints and other areas of the body by using strong magnetic fields and radio frequency pulses to create images of structures inside the body. Worldwide, it is estimated that approximately 60 million MRI procedures are performed each year, according to figures cited by Medtronic.
When programmed to appropriate settings, MR-conditional Medtronic DBS systems allow patients to continue to receive therapy during MRI scans. Previously, patients receiving an MRI scan had their DBS systems turned off before the scan.
“MRI is commonly the method of choice to image the body to diagnose disease or monitor existing conditions, but MRI use has often been limited in patients receiving DBS therapy,” said John Thornton, medical physicist at the National Hospital for Neurology and Neurosurgery in London, the United Kingdom. “Patients receiving DBS therapy can now receive more advantages of MRI technology.”
“Most patients who we consider for a DBS implant have other conditions which may require MRI,” said Ludvic Zrinzo, neurosurgeon at the National Hospital for Neurology and Neurosurgery in London. “The MR Conditional Activa systems mean patients can receive DBS care, and still may have the option of MRI when needed to manage other conditions.”
To receive approval for MR conditional DBS systems, Medtronic developed proprietary test and measurement systems, in conjunction with advanced electromagnetic modeling tools. Existing Activa DBS systems were tested and evaluated across millions of simulated patient scans spanning more than 38,000 unique implant conditions to demonstrate patient safety.
DBS therapy uses a surgically implanted medical device, similar to a pacemaker, to deliver mild electrical pulses to precisely targeted areas of the brain. The stimulation can be programmed and adjusted noninvasively by a trained clinician to maximize symptom control and minimize side effects. More than 125,000 patients worldwide have received Medtronic DBS Therapy. The therapy currently is approved in many locations around the world, including Europe and the United States, for the treatment of the disabling symptoms of essential tremor, advanced Parkinson’s disease and chronic intractable primary dystonia, for which approval in the United States is under a humanitarian device exemption (HDE). In Europe, Canada and Australia, DBS therapy is approved for the treatment of refractory epilepsy. DBS therapy also is approved for the treatment of severe, treatment-resistant obsessive-compulsive disorder in the European Union and Australia, and in the United States under an HDE.
Medtronic plc is headquartered in Dublin, Ireland.