07.22.14
St. Paul, Minn.-based medical device giant St. Jude Medical Inc. has resolved a U.S. Food and Drug Administration (FDA) warning letter it received a year ago.
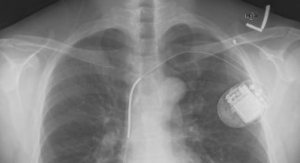
The letter was issued after an inspection of a St. Jude plant in California that manufactures ICD (implantable cardioverter defibrillator) leads (the tiny insulated wires that connect the heart to the implantable device) turned up violations that the company did not address to the FDA’s satisfaction.
In addition to numerous manufacturing, testing and documentation deficiencies, the letter stated that the company failed to report within 30 days malfunctions relating to Durata leads that “would be likely to cause or contribute to a death or serious injury, if the malfunction were to recur.”
“We take our responsibility as a medical device manufacturer very seriously. We are encouraged by the resolution of the FDA’s warning letter and will continue to work to ensure the highest standards are met across our manufacturing facilities,” said Chairman, President and CEO Daniel Starks. “St. Jude Medical is committed to providing our patients, customers and regulators with products and services that meet or exceed their expectations of safety, high-quality performance, reliability and service.”
Over the past few years, St. Jude has had issues—warning letters, negative press, recalls, etc.—regarding its leads. In late 2010, the company shelved Riata defibrillator leads after an Irish study showed wires came through the lead’s outer coating. In December of 2011, the FDA issued a recall of the Riata line.
Taking the Lead (Away)
One way to solve the problems with leads is to innovate them out of the picture. St. Jude Medical’s Nanostim leadless pacemaker recently received an international award—the 2014 Innovation Award at Cardiostim 2014, the 19th World Congress in Cardiac Electrophysiology and Cardiac Techniques held in Nice, France. The award was chosen by a congress committee of electrophysiologists.
More than 4 million people worldwide have an implanted pacemaker or other cardiac rhythm management device, and an additional 700,000 patients receive the devices each year. Unlike conventional pacemakers that require more invasive surgery, the Nanostim is designed to be implanted directly into the heart via a non-surgical procedure. The device, which earned European CE mark in 2013, is placed without the visible surgical pocket, scar and leads required for conventional pacemakers. Implanted via the femoral vein (in the leg) with a tiny delivery system, Nanostim is designed to be fully retrievable so that it can be readily repositioned throughout the implant procedure and later, if necessary. Nanostim is less than 10 percent the size of a conventional pacemaker. The small size of the device and lack of a surgical pocket, coupled with the exclusion of a lead, improves patient comfort and can reduce traditional pacemaker complications, including device pocket-related infection and lead failure, St. Jude Medical officials claim.
Cardiac pacemakers are used to treat bradycardia, which is a heart rate that is too slow. These devices monitor the heart and provide electrical stimulation when the heart beats too slowly for each patient’s specific physiological requirements.
St. Jude Medical has four major clinical focus areas: cardiac rhythm management, atrial fibrillation, cardiovascular and neuromodulation.
The letter was issued after an inspection of a St. Jude plant in California that manufactures ICD (implantable cardioverter defibrillator) leads (the tiny insulated wires that connect the heart to the implantable device) turned up violations that the company did not address to the FDA’s satisfaction.
In addition to numerous manufacturing, testing and documentation deficiencies, the letter stated that the company failed to report within 30 days malfunctions relating to Durata leads that “would be likely to cause or contribute to a death or serious injury, if the malfunction were to recur.”
“We take our responsibility as a medical device manufacturer very seriously. We are encouraged by the resolution of the FDA’s warning letter and will continue to work to ensure the highest standards are met across our manufacturing facilities,” said Chairman, President and CEO Daniel Starks. “St. Jude Medical is committed to providing our patients, customers and regulators with products and services that meet or exceed their expectations of safety, high-quality performance, reliability and service.”
Over the past few years, St. Jude has had issues—warning letters, negative press, recalls, etc.—regarding its leads. In late 2010, the company shelved Riata defibrillator leads after an Irish study showed wires came through the lead’s outer coating. In December of 2011, the FDA issued a recall of the Riata line.
Taking the Lead (Away)
One way to solve the problems with leads is to innovate them out of the picture. St. Jude Medical’s Nanostim leadless pacemaker recently received an international award—the 2014 Innovation Award at Cardiostim 2014, the 19th World Congress in Cardiac Electrophysiology and Cardiac Techniques held in Nice, France. The award was chosen by a congress committee of electrophysiologists.
More than 4 million people worldwide have an implanted pacemaker or other cardiac rhythm management device, and an additional 700,000 patients receive the devices each year. Unlike conventional pacemakers that require more invasive surgery, the Nanostim is designed to be implanted directly into the heart via a non-surgical procedure. The device, which earned European CE mark in 2013, is placed without the visible surgical pocket, scar and leads required for conventional pacemakers. Implanted via the femoral vein (in the leg) with a tiny delivery system, Nanostim is designed to be fully retrievable so that it can be readily repositioned throughout the implant procedure and later, if necessary. Nanostim is less than 10 percent the size of a conventional pacemaker. The small size of the device and lack of a surgical pocket, coupled with the exclusion of a lead, improves patient comfort and can reduce traditional pacemaker complications, including device pocket-related infection and lead failure, St. Jude Medical officials claim.
Cardiac pacemakers are used to treat bradycardia, which is a heart rate that is too slow. These devices monitor the heart and provide electrical stimulation when the heart beats too slowly for each patient’s specific physiological requirements.
St. Jude Medical has four major clinical focus areas: cardiac rhythm management, atrial fibrillation, cardiovascular and neuromodulation.