04.08.14
Medtronic Inc. has enrolled the first patients in the Improve SCA clinical study, a first-of-its-kind trial designed to identify patients in developing countries at a high risk for sudden cardiac arrest (SCA).
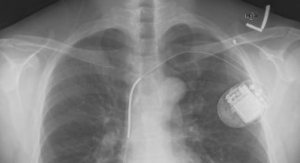
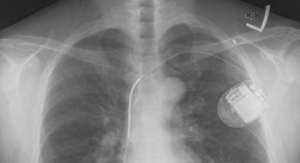
The company enrolled its first patients in the Improve SCA clinical study at West China Hospital and Fuwai Hospital in China. These patients may be indicated for either an implantable cardioverter defibrillator (ICD) or a cardiac resynchronization therapy defibrillator (CRT-D).
ICD administers electrical shocks or painless pacing therapy to stop the heart from quivering chaotically, while CRT-D resynchronizes the contractions of the ventricles by sending tiny electrical impulses to the heart.
The two devices stop a life-threatening fast or irregular heart rhythm that causes SCA. The devices are under-used in developing countries with typically less than 10 percent of indicated patients implanted.
The prospective, non-randomized, multicenter study will build local clinical evidence to identify already indicated ICD/CRT-D primary prevention patients with additional risk factors that put them at a highest risk for sudden cardiac arrest (SCA).
The additional risk factors include fainting (syncope), rapid transient abnormal heartbeats starting in the bottom chambers of the heart (non-sustained ventricular tachycardia or frequent premature ventricular contractions), or, decreased function of the heart (low ejection fraction).
Medtronic’s Improve SCA study will be conducted at approximately 100 sites in more than 15 countries across Central and Eastern Europe, Asia, Middle East and Africa, India, and Latin America. The study is expected to enroll approximately 4,800 participants that will be followed for two years.
The study will evaluate patients meeting current Class I guidelines for ICD (or CRT-D) device implants. It will test whether patients with a primary prevention indication for an ICD and one or more additional risk factors have the same risk of life-threatening ventricular arrhythmia as patients who have survived a prior episode of life-threatening ventricular arrhythmia and are indicated for an ICD for secondary prevention of SCA.
Medtronic said the study results may help clinicians identify and refer primary prevention patients for ICD/CRT-D therapy, and help more patients make informed decisions about receiving a defibrillator.
The company enrolled its first patients in the Improve SCA clinical study at West China Hospital and Fuwai Hospital in China. These patients may be indicated for either an implantable cardioverter defibrillator (ICD) or a cardiac resynchronization therapy defibrillator (CRT-D).
ICD administers electrical shocks or painless pacing therapy to stop the heart from quivering chaotically, while CRT-D resynchronizes the contractions of the ventricles by sending tiny electrical impulses to the heart.
The two devices stop a life-threatening fast or irregular heart rhythm that causes SCA. The devices are under-used in developing countries with typically less than 10 percent of indicated patients implanted.
The prospective, non-randomized, multicenter study will build local clinical evidence to identify already indicated ICD/CRT-D primary prevention patients with additional risk factors that put them at a highest risk for sudden cardiac arrest (SCA).
The additional risk factors include fainting (syncope), rapid transient abnormal heartbeats starting in the bottom chambers of the heart (non-sustained ventricular tachycardia or frequent premature ventricular contractions), or, decreased function of the heart (low ejection fraction).
Medtronic’s Improve SCA study will be conducted at approximately 100 sites in more than 15 countries across Central and Eastern Europe, Asia, Middle East and Africa, India, and Latin America. The study is expected to enroll approximately 4,800 participants that will be followed for two years.
The study will evaluate patients meeting current Class I guidelines for ICD (or CRT-D) device implants. It will test whether patients with a primary prevention indication for an ICD and one or more additional risk factors have the same risk of life-threatening ventricular arrhythmia as patients who have survived a prior episode of life-threatening ventricular arrhythmia and are indicated for an ICD for secondary prevention of SCA.
Medtronic said the study results may help clinicians identify and refer primary prevention patients for ICD/CRT-D therapy, and help more patients make informed decisions about receiving a defibrillator.