03.13.14
Johnson & Johnson’s Cordis subsidiary has obtained the European CE mark for its Renlane renal denervation system to treat high blood pressure. Moving the company product line forward with what has become a slightly controversial technology. Earlier this year, Medtronic Inc. suspended a high-profile clinical trial for its renal denervation device in the United States based on surprising study results (though the technology already is approved and sold in Europe). And Covidien plc announced plans to exit the market. Companies such as Boston Scientific Corp. are still evaluating. Some start-ups are moving ahead full bore.
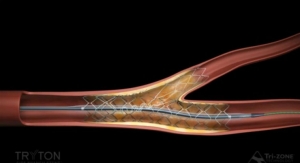
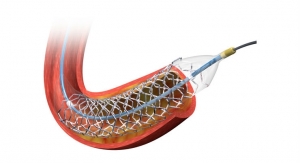
The Renlane renal denervation catheter features five irrigated electrodes located at the tip of the ablation catheter and is used in conjunction with the Renlane Multi-Channel radiofrequency (RF) generator for energy delivery. The system—which includes a helical shaped, irrigated, multi-electrode ablation catheter with a multi-channel radiofrequency ablation system—is used to denervate the renal arteries to reduce blood pressure in patients suffering from drug-resistant hypertension.
The minimally invasive procedure involves applying RF ablation through a catheter to the renal arteries that supply the kidneys with blood, causing the kidneys to produce less of the hormones that cause chronic high blood pressure.
The World Health Organization estimates 1 billion people globally live with hypertension or high blood pressure, and the disease is the cause of one in every eight deaths. Hypertension currently is the third leading cause of death worldwide.
Roughly 15 percent to 30 percent of treated hypertensive patients are resistant to traditional drug therapy, defined as failure to respond to three or more drugs. The International Society of Hypertension estimates the annual cost of healthcare expenditure directly related to elevated blood pressure to be almost $500 billion.
Cordis worldwide president Celine Martin said, “We are pleased to make our Renlane renal denervation system available to European clinicians in need of solutions for patients who do not respond to traditional drug therapy. We are looking forward to gaining more experience with this therapy and making it available to more patients in need of treatment around the world.”
The Renlane renal denervation catheter features five irrigated electrodes located at the tip of the ablation catheter and is used in conjunction with the Renlane Multi-Channel radiofrequency (RF) generator for energy delivery. The system—which includes a helical shaped, irrigated, multi-electrode ablation catheter with a multi-channel radiofrequency ablation system—is used to denervate the renal arteries to reduce blood pressure in patients suffering from drug-resistant hypertension.
The minimally invasive procedure involves applying RF ablation through a catheter to the renal arteries that supply the kidneys with blood, causing the kidneys to produce less of the hormones that cause chronic high blood pressure.
The World Health Organization estimates 1 billion people globally live with hypertension or high blood pressure, and the disease is the cause of one in every eight deaths. Hypertension currently is the third leading cause of death worldwide.
Roughly 15 percent to 30 percent of treated hypertensive patients are resistant to traditional drug therapy, defined as failure to respond to three or more drugs. The International Society of Hypertension estimates the annual cost of healthcare expenditure directly related to elevated blood pressure to be almost $500 billion.
Cordis worldwide president Celine Martin said, “We are pleased to make our Renlane renal denervation system available to European clinicians in need of solutions for patients who do not respond to traditional drug therapy. We are looking forward to gaining more experience with this therapy and making it available to more patients in need of treatment around the world.”