Francisco Almada Lobo , Contributing Writer02.14.14
The way most manufacturers see quality is greatly influenced by Dr. W. Edwards Deming, Ph.D., best known as the father of the “plan, do, check, act” cycle. Thinking of manufacturing as a holistic system and applying such an approach to quality often is easier said than done.
This article will provide a foundation for the argument for why the best platform for implementing a quality system is the manufacturing execution system or manufacturing operations management (MES/MOM) platform.
Most medical device companies are growing rapidly, but only about a quarter of them are growing and improving their financial results at the same time. Figure 1 shows that medical device manufacturers who have achieved both (the “Advancers”) are much more likely to use software applications than other medical device makers.1 In support of our argument, two of the applications where the disparity in use is largest are MES/MOM and operations or manufacturing intelligence (OI/MI).
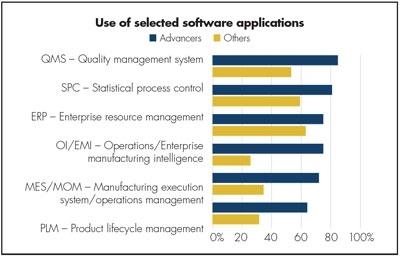
Figure 1. Advancers that had significant organic growth and improvements in financial performance are far more likely to use software, and particularly plant-floor MES/MOM and OI/MI.
Reason Number One: Quality is (or Should Be) Everyone’s Daily Business
One of Deming’s most famous quotes is: “Quality is everyone’s responsibility.” The consequence for manufacturing systems is straightforward: Quality must involve everyone in manufacturing, from the shop floor to the top floor. The quality processes in manufacturing must be fully embedded in the general culture and manufacturing processes. A major source of future discrepancies, even if unintentional, is the creation of two subsystems, one for manufacturing operations and another for quality operations.
When a production operator reports a deviation, triggers a quality protocol, executes a correction or containment action as part of a corrective and preventive action (CAPA), he or she can have no doubts about which system to use to achieve this goal. This is the primary reason why MES/MOM, the broader manufacturing system, is the best platform to encompass manufacturing quality management operations.
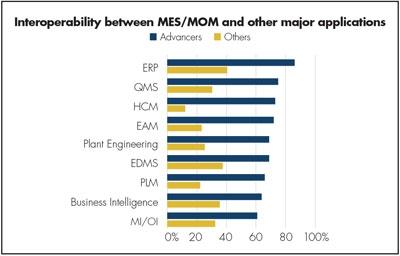
Figure 2. More of the successful Advancer medical device companies have information flows among MES/MOM and all their other enterprise applications.
The following nine reasons all are consequences of this strategic approach.
2. Information Flow
Enforcing quality standards is becoming more complicated. As products are more complex and differentiated, processes simultaneously need to be flexible and robust. One of the challenges is that the process is multi-disciplinary, yet the users should not have to know that it’s working in different sub-systems. The different sub-systems need to have the ability to work together seamlessly.
So it comes as no surprise that most Advancers have information flow from and to the MES/MOM platform. Figure 2 shows this, and note that the difference particularly is strong with the quality management system (QMS), right after enterprise resource planning.
The need for this flow is easy to demonstrate. Consider a scenario where a manufacturing operator moves a lot out of an operation. This lot/material collects engineering data from the equipment, calculates derived parameters or indicators in real time, associates them with the lot in electronic data collection, and posts the data into a statistical process control (SPC) chart. If it detects a deviation, the system can hold the lot in question and trigger an automatic CAPA protocol.
This is a typical manufacturing workflow used often in many discrete industries. If MES/MOM and quality management are disparate systems, interoperability is still possible. But it implies a tremendous effort for the different scenarios. And far from being a one-time effort—such integration needs to be monitored, maintained and evolved.
3. Master Data
If the subsystems, despite being interoperable, belong to different platforms, one of the most difficult aspects (which often is ignored), is the master data (i.e., non-transactional data entities). The master data may include flow, step, resources, states or parameters, and other information that manufacturing and quality will need to be effective.
The master data elements are necessary for an MES transaction (moving a lot into an operation, for example) as well as for a quality functions (such as CAPA, incoming material inspections or operator quality instructions). If the systems are separated, there are only two possibilities: Master data is duplicated, or it is synchronized from an external master data management system.
A master data system includes a set of processes, governance and tools to consistently define and manage the master data. The effort associated with removing duplicates, removing incorrect entered data and overall data synchronization and standardization can become huge.
If all these functions and features belong to the same platform, the master data can be unique, created and managed within the same system, saving significant time and effort, not to mention the elimination of errors caused by wrong master data.
4. Electronic Device History Records
The U.S. Food and Drug Administration’s CFR Title 21 requires manufacturers to maintain device history records (DHR) for each batch or lot, to demonstrate it has been built in accordance with its device master record, and must include all dates, quantities, acceptance records, labels and device identifiers along the manufacturing steps.
DHRs traditionally have been maintained by paper records, including production travelers and other documentation collected into the final record. Its electronic paperless version, the eDHR, when implemented in the same MES/MOM platform, automatically captures all required information in real-time and maintains an always accessible complete set of DHRs for every unit in the production line. In fact, the DHR doesn’t even need to be built, as it represents a view of the complete history records maintained by MES systems for quality related purposes.
Furthermore, this same eDHR implemented in the MES allows immediate navigation into related reports or more detailed information from within the same system. So beyond regulatory compliance, this approach allows visibility and traceability into manufacturing operations. In this environment, problem resolution practices can be implemented easily, effectively and quickly as the MES provides real-time information and guidance.
5. Additional Regulatory Compliance
Beyond the eDHR, several other CFR21 requirements are significantly easier to comply with if implemented in a common shop-floor platform. This includes system validations, audit trails, electronic signatures and documentation.
A few examples include:
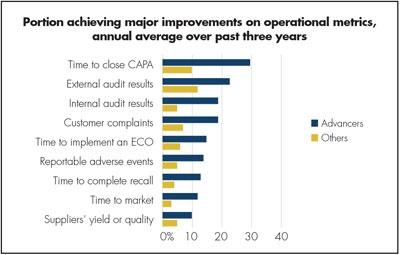
Figure 3. Advancers are far more likely to have improved time to close a CAPA than others. Remember, they also are more likely to have quality and MES/MOM integrated.
6. Integrated CAPA
In the same study, Advancers have achieved major improvements during a three-year period related to the time to close a CAPA as shown in Figure 3. The time to close a CAPA can significantly be shortened with an integrated CAPA system. First of all, such a system allows workflow real-time triggering from shop-floor data such as material parameters, yields or loss types, engineering parameter levels, statistical process control violation, equipment alarm or events. Then the integrated system provides all the necessary data for the affected products or equipment, reducing the time required to search for information.
One essential step of any problem-solving methodology is containment. Containment does not eliminate the root cause of the problem, but instead tries to limit it. Prompt action is extremely important because the effects of the detected problem must not develop further and, above all, defects must not reach the customer.
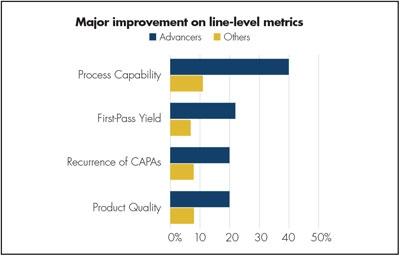
Figure 4. Improvements on the production line are a foundation for better business performance, and the correlation is clear here as Advancers are several times more likely to have improved on important quality metrics.
The implementation of CAPA within the MES/MOM platform then has the advantage of allowing an easy search and subsequent action on material or equipment under investigation. This is especially important as this search might not be trivial. For example, one might find that a given parameter in a specific piece of equipment is causing a faulty behavior of an end-product.
A containment action might include:
7. Process Control
SPC is a very effective method of monitoring a manufacturing process through the use of control charts, upon collecting in-process data. Since Deming introduced these quality control methods, these have been considered a core element of any quality management system. Process capability is the number-one improvement in line-level metrics for Advancers as shown in Figure 4, and far more of them have made major improvements in this metric.
If the SPC system is not integrated with the MES/MOM platform, it will become extremely difficult to successfully monitor and control process input variables and therefore control the process variability. Measuring output only creates alarm signals that are too late to prevent problems or improve the outcomes of that process.
Moreover, upon deviations, several actions might be triggered, such as the opening of quality deviation protocols. But immediate containment actions are necessary such as holding the material or the equipment, or restricting the material to certain operations (e.g., prevent lot merges or prevent lot shipments while the deviation causes and consequences are being investigated).
Both the online SPC with in-process collected data and the immediate reaction and containment actions are key. This favors the usage of a common platform, as opposed to a separate SPC or quality management system. In the more sophisticated advanced process control, the need to incorporate feedback or feed-forward variables in real-time makes the need for a common platform even more obvious.
8. Manufacturing Document Control
Documentation control is an essential quality management function. Most manufacturing processes involve constant collaboration amongst several departments as products and related product documentation are manufactured and written. Training documents, work instructions, standard operating procedures or any other type of manufacturing-related documents have the common life-cycle requirements of any other document. They need to be created, versioned, reviewed, changed, approved, published or deactivated. Therefore, they could be, in principle, managed by standard document control repositories that use version control to track document versions, approvals and notifications.
However, the document management system (DMS) can be taken to a completely different level when integrated into the MES platform. It then becomes capable of grasping the context in which a particular document is being used. From sourcing the raw material to executing each stage of production and finally shipping the finished goods, the display of work instructions or production plans at every stage of the production cycle until the final product is manufactured is crucial.
But even before this happens, the documents have gone through a custom approval process, tailored to the area and function. For instance, a work instruction has probably gone through approval by product engineering, quality, process and operations departments before being deployed to a given context. This manufacturing operations context for creation, approval and visualization of documents can never be so effectively implemented if the document control system is not smoothly integrated into the MES/MOM platform.
9. Equipment Management
Quality management systems require equipment management modules to comply with process control requirements (equipment preventive maintenance) and control of inspection and testing equipment (calibration management). These modules are designed to allow easy scheduling and recording of all calibration and maintenance activities, but these features have obvious advantages when integrated with the overall equipment management and scheduling, as well as with CAPA, which can both be in MES. There are several advantages to the MES approach.
On one side, preventive maintenance and calibration activities can be done based on real usage instead of just time-based. On the other, they can be planned in conjunction with other productive activities of the equipment, allowing re-scheduling in case of deviations. Quite importantly, the system automatically will prevent equipment from being productively used in case calibration or maintenance is past due.
Moreover, it becomes possible to use the same MES system for showing or guiding the user through the detailed calibration and/or preventive maintenance instructions. All the while, the system is recording all actions performed and results obtained, for online access and auditing at any time.
10. Single Source of Truth for Reporting and Analysis
According to LNS Research, 55 percent of executives say their quality metrics are not effectively measured and 47 percent say their organizations have too many disparate quality systems and data sources.2 This comes as no surprise with quality systems being separated from the manufacturing operations software, and sometimes not even grouped in the same platform for quality related functions.
A single source of truth requires structuring information models in which every data element is stored exactly once. Consequently, when any such data element is updated, this update propagates to the upper aggregated levels, without duplication or update possibilities.
This means that quality indicators at different levels—typically online, operational data store and data warehouse—must be based on the same quality and manufacturing operations facts: the single source of true manufacturing data. These indicators then need additional source, transformations and aggregations while moving up the levels. This is the only way a full range of quality related actions, from the lower-level shop-floor to upper level management strategies, can be performed upon the same facts.
* * *
Medical device manufacturers should consider using MES/MOM as the main quality system for these ten reasons. It helps make quality everyone’s business while making it more systematic. It helps consistent data and master data to flow where it’s needed, when it’s needed. Integrated MES/QMS allows eDHR to be not only ready for regulators, but ready to analyze for manufacturing improvements. Built-in integration facilitates and speeds CAPA, process control and equipment management as well as document control processes. As opportunities, regulations and product lines continue to shift, being able to access all manufacturing and quality data electronically is the foundation for analysis and making good decisions as well.
Advancers are doing quite a few things right, and one of them is having integrated MES/MOM and quality information systems in a unified platform. If you do not have an integrated MES/Quality system today, consider what you might be missing and how much it is costing you.
References:
Francisco Almada Lobo is CEO of Porto, Portugal-based Critical Manufacturing, which creates software solutions for advanced manufacturing industries. Prior to Critical, he managed MES/MOM at Siemens, Infineon and Qimonda. He holds an MBA from Escola de Gestão do Porto in Portugal. He can be reached at francisco.lobo@criticalmanufacturing.com or +351 917784754.
This article will provide a foundation for the argument for why the best platform for implementing a quality system is the manufacturing execution system or manufacturing operations management (MES/MOM) platform.
Most medical device companies are growing rapidly, but only about a quarter of them are growing and improving their financial results at the same time. Figure 1 shows that medical device manufacturers who have achieved both (the “Advancers”) are much more likely to use software applications than other medical device makers.1 In support of our argument, two of the applications where the disparity in use is largest are MES/MOM and operations or manufacturing intelligence (OI/MI).
Figure 1. Advancers that had significant organic growth and improvements in financial performance are far more likely to use software, and particularly plant-floor MES/MOM and OI/MI.
One of Deming’s most famous quotes is: “Quality is everyone’s responsibility.” The consequence for manufacturing systems is straightforward: Quality must involve everyone in manufacturing, from the shop floor to the top floor. The quality processes in manufacturing must be fully embedded in the general culture and manufacturing processes. A major source of future discrepancies, even if unintentional, is the creation of two subsystems, one for manufacturing operations and another for quality operations.
When a production operator reports a deviation, triggers a quality protocol, executes a correction or containment action as part of a corrective and preventive action (CAPA), he or she can have no doubts about which system to use to achieve this goal. This is the primary reason why MES/MOM, the broader manufacturing system, is the best platform to encompass manufacturing quality management operations.
Figure 2. More of the successful Advancer medical device companies have information flows among MES/MOM and all their other enterprise applications.
2. Information Flow
Enforcing quality standards is becoming more complicated. As products are more complex and differentiated, processes simultaneously need to be flexible and robust. One of the challenges is that the process is multi-disciplinary, yet the users should not have to know that it’s working in different sub-systems. The different sub-systems need to have the ability to work together seamlessly.
So it comes as no surprise that most Advancers have information flow from and to the MES/MOM platform. Figure 2 shows this, and note that the difference particularly is strong with the quality management system (QMS), right after enterprise resource planning.
The need for this flow is easy to demonstrate. Consider a scenario where a manufacturing operator moves a lot out of an operation. This lot/material collects engineering data from the equipment, calculates derived parameters or indicators in real time, associates them with the lot in electronic data collection, and posts the data into a statistical process control (SPC) chart. If it detects a deviation, the system can hold the lot in question and trigger an automatic CAPA protocol.
This is a typical manufacturing workflow used often in many discrete industries. If MES/MOM and quality management are disparate systems, interoperability is still possible. But it implies a tremendous effort for the different scenarios. And far from being a one-time effort—such integration needs to be monitored, maintained and evolved.
3. Master Data
If the subsystems, despite being interoperable, belong to different platforms, one of the most difficult aspects (which often is ignored), is the master data (i.e., non-transactional data entities). The master data may include flow, step, resources, states or parameters, and other information that manufacturing and quality will need to be effective.
The master data elements are necessary for an MES transaction (moving a lot into an operation, for example) as well as for a quality functions (such as CAPA, incoming material inspections or operator quality instructions). If the systems are separated, there are only two possibilities: Master data is duplicated, or it is synchronized from an external master data management system.
A master data system includes a set of processes, governance and tools to consistently define and manage the master data. The effort associated with removing duplicates, removing incorrect entered data and overall data synchronization and standardization can become huge.
If all these functions and features belong to the same platform, the master data can be unique, created and managed within the same system, saving significant time and effort, not to mention the elimination of errors caused by wrong master data.
4. Electronic Device History Records
The U.S. Food and Drug Administration’s CFR Title 21 requires manufacturers to maintain device history records (DHR) for each batch or lot, to demonstrate it has been built in accordance with its device master record, and must include all dates, quantities, acceptance records, labels and device identifiers along the manufacturing steps.
DHRs traditionally have been maintained by paper records, including production travelers and other documentation collected into the final record. Its electronic paperless version, the eDHR, when implemented in the same MES/MOM platform, automatically captures all required information in real-time and maintains an always accessible complete set of DHRs for every unit in the production line. In fact, the DHR doesn’t even need to be built, as it represents a view of the complete history records maintained by MES systems for quality related purposes.
Furthermore, this same eDHR implemented in the MES allows immediate navigation into related reports or more detailed information from within the same system. So beyond regulatory compliance, this approach allows visibility and traceability into manufacturing operations. In this environment, problem resolution practices can be implemented easily, effectively and quickly as the MES provides real-time information and guidance.
5. Additional Regulatory Compliance
Beyond the eDHR, several other CFR21 requirements are significantly easier to comply with if implemented in a common shop-floor platform. This includes system validations, audit trails, electronic signatures and documentation.
A few examples include:
- Training requirements can be enforced through MOM certification modules, which automatically allow only certified personnel to perform certain types of transactions at given steps and for given products.
- Certain tasks require confirmation with signatures from two different persons or roles, which can be implemented in the MES system with the associated electronic records and audit trails.
- Several types of validity checks can be implemented to determine the validity of the source of data input or operational instruction.
- Labeling requirements also can be implemented easily in modern MOM platforms, particularly important given the specific dynamic shop-floor context required to print the right information on labels.
Figure 3. Advancers are far more likely to have improved time to close a CAPA than others. Remember, they also are more likely to have quality and MES/MOM integrated.
In the same study, Advancers have achieved major improvements during a three-year period related to the time to close a CAPA as shown in Figure 3. The time to close a CAPA can significantly be shortened with an integrated CAPA system. First of all, such a system allows workflow real-time triggering from shop-floor data such as material parameters, yields or loss types, engineering parameter levels, statistical process control violation, equipment alarm or events. Then the integrated system provides all the necessary data for the affected products or equipment, reducing the time required to search for information.
One essential step of any problem-solving methodology is containment. Containment does not eliminate the root cause of the problem, but instead tries to limit it. Prompt action is extremely important because the effects of the detected problem must not develop further and, above all, defects must not reach the customer.
Figure 4. Improvements on the production line are a foundation for better business performance, and the correlation is clear here as Advancers are several times more likely to have improved on important quality metrics.
The implementation of CAPA within the MES/MOM platform then has the advantage of allowing an easy search and subsequent action on material or equipment under investigation. This is especially important as this search might not be trivial. For example, one might find that a given parameter in a specific piece of equipment is causing a faulty behavior of an end-product.
A containment action might include:
- Preventing the equipment from further processing until it is investigated;
- Searching for all materials processed by this specific equipment;
- Defining either within a certain interval and/or when the mentioned parameter was set above a given threshold value; and
- Holding all of the products that might be involved.
7. Process Control
SPC is a very effective method of monitoring a manufacturing process through the use of control charts, upon collecting in-process data. Since Deming introduced these quality control methods, these have been considered a core element of any quality management system. Process capability is the number-one improvement in line-level metrics for Advancers as shown in Figure 4, and far more of them have made major improvements in this metric.
If the SPC system is not integrated with the MES/MOM platform, it will become extremely difficult to successfully monitor and control process input variables and therefore control the process variability. Measuring output only creates alarm signals that are too late to prevent problems or improve the outcomes of that process.
Moreover, upon deviations, several actions might be triggered, such as the opening of quality deviation protocols. But immediate containment actions are necessary such as holding the material or the equipment, or restricting the material to certain operations (e.g., prevent lot merges or prevent lot shipments while the deviation causes and consequences are being investigated).
Both the online SPC with in-process collected data and the immediate reaction and containment actions are key. This favors the usage of a common platform, as opposed to a separate SPC or quality management system. In the more sophisticated advanced process control, the need to incorporate feedback or feed-forward variables in real-time makes the need for a common platform even more obvious.
8. Manufacturing Document Control
Documentation control is an essential quality management function. Most manufacturing processes involve constant collaboration amongst several departments as products and related product documentation are manufactured and written. Training documents, work instructions, standard operating procedures or any other type of manufacturing-related documents have the common life-cycle requirements of any other document. They need to be created, versioned, reviewed, changed, approved, published or deactivated. Therefore, they could be, in principle, managed by standard document control repositories that use version control to track document versions, approvals and notifications.
However, the document management system (DMS) can be taken to a completely different level when integrated into the MES platform. It then becomes capable of grasping the context in which a particular document is being used. From sourcing the raw material to executing each stage of production and finally shipping the finished goods, the display of work instructions or production plans at every stage of the production cycle until the final product is manufactured is crucial.
But even before this happens, the documents have gone through a custom approval process, tailored to the area and function. For instance, a work instruction has probably gone through approval by product engineering, quality, process and operations departments before being deployed to a given context. This manufacturing operations context for creation, approval and visualization of documents can never be so effectively implemented if the document control system is not smoothly integrated into the MES/MOM platform.
9. Equipment Management
Quality management systems require equipment management modules to comply with process control requirements (equipment preventive maintenance) and control of inspection and testing equipment (calibration management). These modules are designed to allow easy scheduling and recording of all calibration and maintenance activities, but these features have obvious advantages when integrated with the overall equipment management and scheduling, as well as with CAPA, which can both be in MES. There are several advantages to the MES approach.
On one side, preventive maintenance and calibration activities can be done based on real usage instead of just time-based. On the other, they can be planned in conjunction with other productive activities of the equipment, allowing re-scheduling in case of deviations. Quite importantly, the system automatically will prevent equipment from being productively used in case calibration or maintenance is past due.
Moreover, it becomes possible to use the same MES system for showing or guiding the user through the detailed calibration and/or preventive maintenance instructions. All the while, the system is recording all actions performed and results obtained, for online access and auditing at any time.
10. Single Source of Truth for Reporting and Analysis
According to LNS Research, 55 percent of executives say their quality metrics are not effectively measured and 47 percent say their organizations have too many disparate quality systems and data sources.2 This comes as no surprise with quality systems being separated from the manufacturing operations software, and sometimes not even grouped in the same platform for quality related functions.
A single source of truth requires structuring information models in which every data element is stored exactly once. Consequently, when any such data element is updated, this update propagates to the upper aggregated levels, without duplication or update possibilities.
This means that quality indicators at different levels—typically online, operational data store and data warehouse—must be based on the same quality and manufacturing operations facts: the single source of true manufacturing data. These indicators then need additional source, transformations and aggregations while moving up the levels. This is the only way a full range of quality related actions, from the lower-level shop-floor to upper level management strategies, can be performed upon the same facts.
* * *
Medical device manufacturers should consider using MES/MOM as the main quality system for these ten reasons. It helps make quality everyone’s business while making it more systematic. It helps consistent data and master data to flow where it’s needed, when it’s needed. Integrated MES/QMS allows eDHR to be not only ready for regulators, but ready to analyze for manufacturing improvements. Built-in integration facilitates and speeds CAPA, process control and equipment management as well as document control processes. As opportunities, regulations and product lines continue to shift, being able to access all manufacturing and quality data electronically is the foundation for analysis and making good decisions as well.
Advancers are doing quite a few things right, and one of them is having integrated MES/MOM and quality information systems in a unified platform. If you do not have an integrated MES/Quality system today, consider what you might be missing and how much it is costing you.
References:
- Source for all charts: Beyond Tradeoffs in Medical Device Manufacturing: Balancing Innovation, Quality and Compliance While Improving Profit, 2012, Cambashi Inc. and UBM.
- http://blog.lnsresearch.com/blog/bid/183872/5-Steps-to-Justifying-an-Investment-in-Enterprise-Quality-Software-Infographic.
Francisco Almada Lobo is CEO of Porto, Portugal-based Critical Manufacturing, which creates software solutions for advanced manufacturing industries. Prior to Critical, he managed MES/MOM at Siemens, Infineon and Qimonda. He holds an MBA from Escola de Gestão do Porto in Portugal. He can be reached at francisco.lobo@criticalmanufacturing.com or +351 917784754.



























