06.14.13
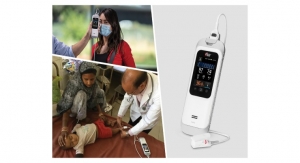
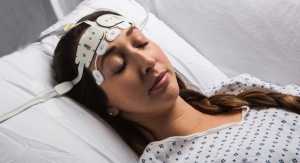
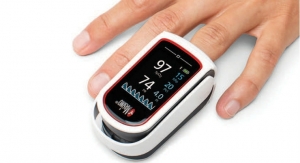
Covidien plc has been granted U.S. Food and Drug Administration (FDA) 510(k) clearance for its Nellcor pulse oximetry portfolio. The line of devices is designed to measure arterial oxygen saturation. According to Covidien, it is the first company to receive clearance for a motion-tolerant bedside pulse oximeter portfolio that also is compliant with ISO 80601-2-61 (the ISO standard for pulse oximetry). Pulse oximeters provide early warning of dangerous respiratory complications, enabling clinicians to detect and address life-threatening events sooner.
The portfolio of devices consists of the Nellcor bedside SpO2 (arterial hemoglobin) patient monitoring system, the Nellcor bedside respiratory patient monitoring system and the Nellcor N-600x pulse oximetry monitoring system.
Nellcor devices rely on cardiac-based signals to provide a more accurate reading that closely is tied to the patient’s physiology. This, Covidien officials claim, drives consistent performance during various challenging conditions, such as patient motion, noise and low perfusion, which can impede the assessment of patient respiratory status. Motion-tolerant pulse oximetry is important for ensuring patient safety because patient movement can thwart accurate readings and delay diagnosis of serious respiratory compromise.
Covidien also offers training in pulse oximetry technology through its new Professional Affairs and Clinical Education Online Platform to support the safe and effective use of motion-tolerant pulse oximeters.
The devices in the newly cleared Nellcor portfolio are indicated for prescription-use only for the continuous non-invasive monitoring of functional oxygen saturation of SpO2 and pulse rate. They are intended for neonatal, pediatric, and adult patients, and for well or poorly perfused patients in hospitals, hospital-type facilities, intra-hospital transport and home environments.
Covidien’s U.S. headquarters are in Mansfield, Mass.
The portfolio of devices consists of the Nellcor bedside SpO2 (arterial hemoglobin) patient monitoring system, the Nellcor bedside respiratory patient monitoring system and the Nellcor N-600x pulse oximetry monitoring system.
|
Covidien Nellcor Pulse Oximetry Bedside Monitors. Image courtesy of Covidien plc. |
Covidien also offers training in pulse oximetry technology through its new Professional Affairs and Clinical Education Online Platform to support the safe and effective use of motion-tolerant pulse oximeters.
The devices in the newly cleared Nellcor portfolio are indicated for prescription-use only for the continuous non-invasive monitoring of functional oxygen saturation of SpO2 and pulse rate. They are intended for neonatal, pediatric, and adult patients, and for well or poorly perfused patients in hospitals, hospital-type facilities, intra-hospital transport and home environments.
Covidien’s U.S. headquarters are in Mansfield, Mass.