05.09.11
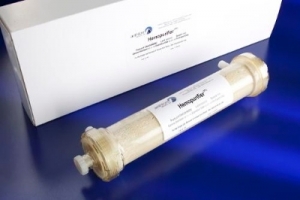
Aethlon Medical Inc. is looking to expand the target treatment indication of its Hemopurifier, a selective filtration device, to include the hepatitis C virus (HCV) in the United States. The company has requested a face-to-face meeting with the U.S. Food and Drug Administration (FDA) to discuss the re-initiation of an Investigational Device Exemption (IDE) study. The current IDE recognizes the use of the Hemopurifier only as a countermeasure against bioterror and pandemic threats.
James Joyce, chairman and CEO of Aethlon, explained that the company’s collection of human safety data has expanded since submitting the original IDE, and that data suggests the Hemopurifier can substantially reduce viral load in HCV-infected patients, even in the absence of antiviral drugs.
“These results combined with an emergence of a remarkable clinical validation for therapeutic filtration in HCV care provide a compelling basis for an FDA meeting to discuss and advance a potential clinical strategy to treat HCV-infected individuals in the United States,” Joyce said.
Aethlon executives believe that the Hemopurifier can be an adjunct to current and future iterations of standard of care (SOC) drug therapy by accelerating viral load depletion in the first few days of HCV-SOC.
Clinical validation for therapeutic filtration of HCV has been demonstrated by the VRAD system, which was developed and marketed by Asahi Kasei Kuraray Medical in Tokyo, Japan. Early administration of VRAD in combination with HCV-SOC drug therapy achieved 71.4 percent sustained virologic response rates (SVR) in HCV patients who previously failed HCV-SOC. This outcome was achieved through a once-daily administration of VRAD for three consecutive days at the outset of SOC therapy. The average viral load reduction during each treatment period was 26.1 percent.
The Hemopurifier demonstrated an average viral load reduction of 64 percent without antiviral drug benefit in a similar treatment timeframe (3 ¼ hours) to that of the VRAD, prompting Aethlon to conduct an adjunct study to show the ability of the Hemopurifier when enhanced with HCV-SOC. The study is being conducted at the Medanta Medicity Institute in Delhi, India.
An additional benefit of the Hemopurifier to HCV patients is its ability to treat those with end-stage renal disease (ESRD), whose HCV-SOC options are typically limited. Treatment with the Hemopurifier involves no drug toxicity or interaction concerns, and can be included with a dialysis cartridge so there is no additional treatment beyond the patient’s pre-established treatment schedule.
Aethlon expects its meeting with the FDA to occur in mid-summer. The company also is planning to start discussions on the necessary regulatory requirements to obtain an Emergency Use Authorization for the Hemopurifier as a broad-spectrum counter-measure against untreatable viral threats that emerge naturally or as an agent of bioterrorism. As the sole antiviral strategy to address drug and vaccine-resistant viral pathogens, Aethlon believes the Hemopurifier is suited to meet the U.S. government’s need for medical countermeasures that can be deployed during national health emergencies.
James Joyce, chairman and CEO of Aethlon, explained that the company’s collection of human safety data has expanded since submitting the original IDE, and that data suggests the Hemopurifier can substantially reduce viral load in HCV-infected patients, even in the absence of antiviral drugs.
“These results combined with an emergence of a remarkable clinical validation for therapeutic filtration in HCV care provide a compelling basis for an FDA meeting to discuss and advance a potential clinical strategy to treat HCV-infected individuals in the United States,” Joyce said.
Aethlon executives believe that the Hemopurifier can be an adjunct to current and future iterations of standard of care (SOC) drug therapy by accelerating viral load depletion in the first few days of HCV-SOC.
Clinical validation for therapeutic filtration of HCV has been demonstrated by the VRAD system, which was developed and marketed by Asahi Kasei Kuraray Medical in Tokyo, Japan. Early administration of VRAD in combination with HCV-SOC drug therapy achieved 71.4 percent sustained virologic response rates (SVR) in HCV patients who previously failed HCV-SOC. This outcome was achieved through a once-daily administration of VRAD for three consecutive days at the outset of SOC therapy. The average viral load reduction during each treatment period was 26.1 percent.
The Hemopurifier demonstrated an average viral load reduction of 64 percent without antiviral drug benefit in a similar treatment timeframe (3 ¼ hours) to that of the VRAD, prompting Aethlon to conduct an adjunct study to show the ability of the Hemopurifier when enhanced with HCV-SOC. The study is being conducted at the Medanta Medicity Institute in Delhi, India.
An additional benefit of the Hemopurifier to HCV patients is its ability to treat those with end-stage renal disease (ESRD), whose HCV-SOC options are typically limited. Treatment with the Hemopurifier involves no drug toxicity or interaction concerns, and can be included with a dialysis cartridge so there is no additional treatment beyond the patient’s pre-established treatment schedule.
Aethlon expects its meeting with the FDA to occur in mid-summer. The company also is planning to start discussions on the necessary regulatory requirements to obtain an Emergency Use Authorization for the Hemopurifier as a broad-spectrum counter-measure against untreatable viral threats that emerge naturally or as an agent of bioterrorism. As the sole antiviral strategy to address drug and vaccine-resistant viral pathogens, Aethlon believes the Hemopurifier is suited to meet the U.S. government’s need for medical countermeasures that can be deployed during national health emergencies.