Sean Fenske, Editor-in-Chief11.13.23
For virtually everyone in the U.S., November marks the beginning of winter, shorter days, decreasing temperatures, and Thanksgiving. For those involved with medical device development and manufacturing, it’s also the month for the Medica and CompaMed trade shows in Dusseldorf, Germany. While last year’s event seemed to bring the crowd’s back following the COVID-induced hiatus, this year may be a more true return to form.
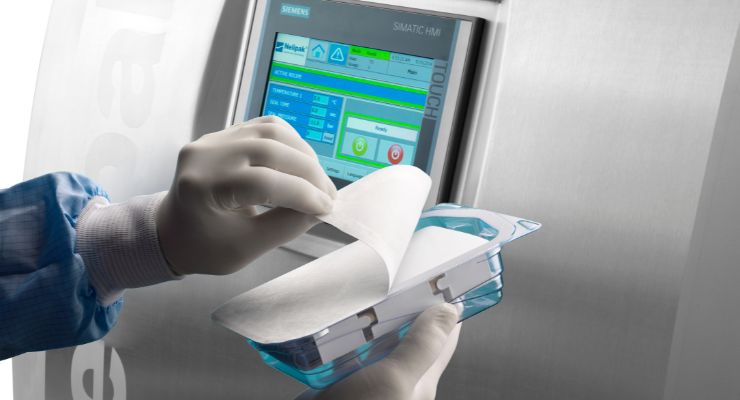
With this in mind, I reached out to a number of companies who will be exhibiting at the show to find out directly from them what they are showing off or focusing on at the event, what challenges customers have brought them, and where they see their role within the industry in aiding medical device manufacturers. Tackling these questions as well as a couple others, Sean Egan, director of global marketing at Nelipak Healthcare Packaging, shared a few insights to help you determine if the firm should be a potential services partner for you in 2024 or beyond.
Sean Fenske: What technology or service are you emphasizing at Medica/CompaMed this year?
Sean Egan: In-silico design is a transformational addition to current design processes proven in medical device and other industries, and it is now being used to enable improvements in packaging. At CompaMed, Nelipak will be emphasizing our NeliSim simulation tool, which is proving very effective in helping customers assess their current and future packaging to understand where opportunities lie to optimize packaging while reducing footprint/material gauge.
Fenske: What’s the most common challenge customers inquire about and how do you address it?
Egan: Sustainability is challenging everyone; not just to reduce waste, but also to be able to recycle product downstream. During CompaMed, Nelipak will present our portfolio of products designed to help customers meet this challenge.
Fenske: If you could give one piece of advice to companies seeking a manufacturing partner before they make a decision, what would it be?
Egan: Ensure a prospective partner truly understands your needs, and has the expertise and competence to deliver and understand the regulatory landscape in which you operate.
Fenske: What are the forces driving medical device manufacturers to seek your technology/services over doing it in-house?
Egan: We cover all aspects of customers’ packaging needs—from design, prototyping, production, validation, to automation—through our expert teams and quality service. This, matched with our footprint, gives customers access to additional services and capabilities not available within their internal teams.
Fenske: In what ways is your company able to aid in getting a product (project) to market faster?
Egan: Nelipak is unique in that we provide both flexible and rigid packaging solutions, backed with validation lab services and equipment all from one supplier. We will be demonstrating this during the show at our booth.
Nelipak Healthcare Packaging is located at Medica/Compamed in Hall 8A, Booth/Stand M01.
With this in mind, I reached out to a number of companies who will be exhibiting at the show to find out directly from them what they are showing off or focusing on at the event, what challenges customers have brought them, and where they see their role within the industry in aiding medical device manufacturers. Tackling these questions as well as a couple others, Sean Egan, director of global marketing at Nelipak Healthcare Packaging, shared a few insights to help you determine if the firm should be a potential services partner for you in 2024 or beyond.
Sean Fenske: What technology or service are you emphasizing at Medica/CompaMed this year?
Sean Egan: In-silico design is a transformational addition to current design processes proven in medical device and other industries, and it is now being used to enable improvements in packaging. At CompaMed, Nelipak will be emphasizing our NeliSim simulation tool, which is proving very effective in helping customers assess their current and future packaging to understand where opportunities lie to optimize packaging while reducing footprint/material gauge.
Fenske: What’s the most common challenge customers inquire about and how do you address it?
Egan: Sustainability is challenging everyone; not just to reduce waste, but also to be able to recycle product downstream. During CompaMed, Nelipak will present our portfolio of products designed to help customers meet this challenge.
Fenske: If you could give one piece of advice to companies seeking a manufacturing partner before they make a decision, what would it be?
Egan: Ensure a prospective partner truly understands your needs, and has the expertise and competence to deliver and understand the regulatory landscape in which you operate.
Fenske: What are the forces driving medical device manufacturers to seek your technology/services over doing it in-house?
Egan: We cover all aspects of customers’ packaging needs—from design, prototyping, production, validation, to automation—through our expert teams and quality service. This, matched with our footprint, gives customers access to additional services and capabilities not available within their internal teams.
Fenske: In what ways is your company able to aid in getting a product (project) to market faster?
Egan: Nelipak is unique in that we provide both flexible and rigid packaging solutions, backed with validation lab services and equipment all from one supplier. We will be demonstrating this during the show at our booth.
Nelipak Healthcare Packaging is located at Medica/Compamed in Hall 8A, Booth/Stand M01.