Jacob Andra, Senior Industry Analyst, U.S. Translation Company09.16.16
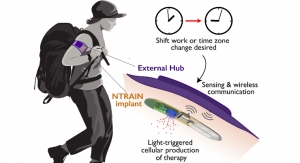
Imagine machines as small as a grain of rice, or even smaller—some invisible to the naked eye. Now, imagine that your body contains these machines, and they are integral to many of your bodily functions. Got rheumatoid arthritis? One of these devices fires electrical impulses into your vagus nerve, easing the effects of your condition. Or, say you’re a soldier on the battlefield; an implant signals your spleen to accelerate blood clotting, stanching the flow from a gunshot wound. Even Parkinson’s and Alzheimer’s may someday be kept in check via brain stimulation from tiny circuits embedded in your gray matter.
Not Just Sci-Fi
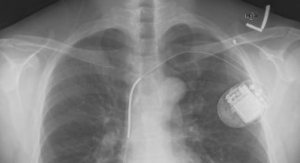
These implants already exist. And, if certain fundamental challenges can be overcome, they’ll be ready for human application in the not-to-distant future. Battelle, for example, plans on bringing their “neural tourniquet” to market by about 2018. They claim that the device can control blood loss, as per the battlefield scenario above. SetPoint Medical has a “microregulator”—a bean-sized implant that will allegedly modulate the immune system (neuromodulation, they call it) to control inflammatory disorders such as Crohn’s disease and rheumatoid arthritis. Several prominent life sciences firms, Boston Scientific among them, have invested in SetPoint.
Further, British pharmaceutical giant GlaxoSmithKline (GSK) has joined forces with Google's life sciences division, Verily. Their joint company, Galvani, will operate independently from both Google and GSK, and will focus solely on bioelectronic medicine. Galvani will explore the role of nerve signals in various health disorders. And how bioelectronic implants can modulate those signals. As the only pharma company aggressively developing bioelectronic implant technologies, GSK is hoping to get far ahead of the pack by the time these tiny implants become mainstream.
Ultimately, these nascent solutions will replace medication, or much of it, providing much more targeted—not to mention effective—treatments, without the side effects. Cancer, diabetes, obesity, asthma, and epilepsy are all targets of bioelectronic medicine, according to Gaia Vince’s Mosiac article, “Bioelectric Dreams.” However, researchers have yet to surmount some formidable obstacles to the realization of bioelectronics treatments. Foremost is the question of how to power the devices.
The Problem of Power
Researchers and engineers already possess most of the constituent technologies needed, but major barriers remain. One of those is figuring out how to power the implants. Since they're designed to remain in small spaces for long periods of time, internal batteries are less than ideal. If you're not going to power the device from inside, the only other option is to power it from the outside. As it turns out, this is really hard to do.
Bioelectronic Disco?
The most promising solution: wireless juicing with "near-infrared-ray (nIR) irradiation." As a Nanowerk article explains, this involves a flashing light that can penetrate into human tissue as deep as 10 cm. The bioelectronic implant absorbs this flashing nIR and experiences temperature fluctuations as a result. Apparently, these rapid changes in temperature produce voltage/current pulses that a team at Xi'an Jiaotong University has used to successfully stimulate the sciatic nerve of a frog, as well as a rat heart.
While the team's stimulation of rodent and amphibian nerves is certainly impressive, one can't help but imagine a future of strobe-lit citizens crowding onto subways or sitting down for dinner in upscale restaurants, their implants gyrating to the rhythm of incoming radiation.
Electricity on a Plate
In another development, a Stanford team sent an electrical current through a "flat plate adorned with a specially designed...conductive material." With this plate—measuring 6 cm on each side—mounted on a hapless rabbit, they powered a rice-sized pacemaker in the bunny's heart. Again, impressive, but more progress is needed. Even if other constraints with the plate-powering system were overcome, the appearance would solicit plenty of objections. Assuming I needed a tiny stimulator in the parietal region of my frontal lobe, I don't know that I'd be thrilled to have a metal square fastened to my forehead. Then again, depending on my ailment, the relief just might justify the Frankensteinian aesthetic. Perhaps such skull plates might spawn a new fashion trend—medi-goth, anyone?
Blood, Sweat, and Tears
Human bodies produce energy; another avenue of research aims to harness it to power devices. We burn 2,000 to 3,000 calories per day; we'd need only to tap a tiny fraction of that to run a small implant.
Turns out, our bodily fluids contain a lot of energy for the taking. Enzymatic biofuel cells (EFC) are already undergoing animal tests for their ability to metabolize energy-rich molecules for electricity generation. Via oxidation, the enzymes in the EFC force electrons into an electrode, which generates a small current. Plasma (the colorless, liquid component of blood) contains dissolved glucose, which many EFCs have been designed to use. Sweat contains lactate, which can power EFCs, but most of us don't sweat continuously. So, that avenue may fizzle. Tears, however, contain a cocktail of energy-rich molecules—and our eyes produce them constantly in minute amounts. Already, bioengineers at the University of Utah have developed an electrode-containing contact lens that "maintained a power output of over one microwatt for three hours" when exposed to a synthetic tear solution.
Kinetic Energy and Piezoelectric Materials
In addition to molecular energy contained in our fluids, we produce kinetic energy whenever we move. Even our internal organs produce enough motion that a group of Chinese and American researchers succeeded in harvesting a microwatt of power from "the beating hearts, lungs, and diaphragms of (sedated) cows and sheep." The mechanism used? Each slumbering beast had an ultra-thin piezoelectric material attached to the respective organ of study.
Piezoelectric (the Greek piezo means to squeeze or press) materials generate a charge when placed under stress. How? Such materials have an atomic lattice structure. Normally, the symmetrical structure of the lattice results in an energetic equilibrium. No difference in potential, thus, no electricity generation. However, when a force deforms the lattice—piezo—a difference in electrical potential emerges—an electrical charge.
In another experiment, researchers lined shoes with an elastomer-based piezoelectric fabric. They powered thirty LED lights from the energy generated by walking. The same group charged a lithium-ion battery by moving a shirt that had piezoelectric fabric applied to it.
Though their application to biomedicine is relatively new, piezoelectric materials have long been used in a number of industrial applications, from optics to motors to robotics. Typically, they reside in a fixed location in crystal form, creating an electrical charge when mechanically stimulated. In addition to their industrial utility, researchers have tried to scale piezoelectric technologies for macro power generation. These efforts have not been successful. Piezoelectric phenomena, it seems, operate under energetic size constraints, which is fine for developers of bioelectronic implants.
Theoretically, then, nothing prevents us from powering our bioelectric implants with hi-tech fabric attached to our organs or piezoelectric clothing that somehow—and here lies the big knowledge gap—transmits electricity to the inside of our bodies. One way or another, piezoelectric materials will almost certainly have prominent applications in the coming bioelectronic revolution.
A Matter of Time
With such attention and resources behind bioelectronic implant development, we can expect even more breakthroughs. Each of the barriers to functional bioelectronic devices—power sources; nerve-implant interfaces; durable, non-degrading materials; sufficiently sophisticated computers within the implants to read and process nerve signals and adjust output accordingly—can be individually solved, given enough research. While these challenges are significant, in the scope of human invention, they're far from insurmountable.
If, in the future, we've got these small medical devices inside of each of us, assisting our bodies with control of function, does that make us cyborgs? Invisible cyborgs, perhaps, because we'd lack the external post-apocalyptic cyborg accouterments of pop film. Does it really matter? Probably not, but it is fun to consider. Philosophical questions abound such as, for example, if we systematically add one piece of electromechanical equipment after another to modify our bodies, at what point do we stop being human and become mechanical? Or are we still human, even after every component of us has been replaced? Fascinating thought experiments. For now, however, if tiny medical devices can signal our nerves and help us reverse neurological disorders, we've accomplished a very good thing.
Jacob Andra is senior industry analyst for U.S. Translation Company, a localization firm that helps life sciences firms localize their products for overseas markets. He can be found on LinkedIn and Twitter.
Note: This article has been revised from a U.S. Translation Company blog post.
Not Just Sci-Fi
These implants already exist. And, if certain fundamental challenges can be overcome, they’ll be ready for human application in the not-to-distant future. Battelle, for example, plans on bringing their “neural tourniquet” to market by about 2018. They claim that the device can control blood loss, as per the battlefield scenario above. SetPoint Medical has a “microregulator”—a bean-sized implant that will allegedly modulate the immune system (neuromodulation, they call it) to control inflammatory disorders such as Crohn’s disease and rheumatoid arthritis. Several prominent life sciences firms, Boston Scientific among them, have invested in SetPoint.
Further, British pharmaceutical giant GlaxoSmithKline (GSK) has joined forces with Google's life sciences division, Verily. Their joint company, Galvani, will operate independently from both Google and GSK, and will focus solely on bioelectronic medicine. Galvani will explore the role of nerve signals in various health disorders. And how bioelectronic implants can modulate those signals. As the only pharma company aggressively developing bioelectronic implant technologies, GSK is hoping to get far ahead of the pack by the time these tiny implants become mainstream.
Ultimately, these nascent solutions will replace medication, or much of it, providing much more targeted—not to mention effective—treatments, without the side effects. Cancer, diabetes, obesity, asthma, and epilepsy are all targets of bioelectronic medicine, according to Gaia Vince’s Mosiac article, “Bioelectric Dreams.” However, researchers have yet to surmount some formidable obstacles to the realization of bioelectronics treatments. Foremost is the question of how to power the devices.
The Problem of Power
Researchers and engineers already possess most of the constituent technologies needed, but major barriers remain. One of those is figuring out how to power the implants. Since they're designed to remain in small spaces for long periods of time, internal batteries are less than ideal. If you're not going to power the device from inside, the only other option is to power it from the outside. As it turns out, this is really hard to do.
Bioelectronic Disco?
The most promising solution: wireless juicing with "near-infrared-ray (nIR) irradiation." As a Nanowerk article explains, this involves a flashing light that can penetrate into human tissue as deep as 10 cm. The bioelectronic implant absorbs this flashing nIR and experiences temperature fluctuations as a result. Apparently, these rapid changes in temperature produce voltage/current pulses that a team at Xi'an Jiaotong University has used to successfully stimulate the sciatic nerve of a frog, as well as a rat heart.
While the team's stimulation of rodent and amphibian nerves is certainly impressive, one can't help but imagine a future of strobe-lit citizens crowding onto subways or sitting down for dinner in upscale restaurants, their implants gyrating to the rhythm of incoming radiation.
Electricity on a Plate
In another development, a Stanford team sent an electrical current through a "flat plate adorned with a specially designed...conductive material." With this plate—measuring 6 cm on each side—mounted on a hapless rabbit, they powered a rice-sized pacemaker in the bunny's heart. Again, impressive, but more progress is needed. Even if other constraints with the plate-powering system were overcome, the appearance would solicit plenty of objections. Assuming I needed a tiny stimulator in the parietal region of my frontal lobe, I don't know that I'd be thrilled to have a metal square fastened to my forehead. Then again, depending on my ailment, the relief just might justify the Frankensteinian aesthetic. Perhaps such skull plates might spawn a new fashion trend—medi-goth, anyone?
Blood, Sweat, and Tears
Human bodies produce energy; another avenue of research aims to harness it to power devices. We burn 2,000 to 3,000 calories per day; we'd need only to tap a tiny fraction of that to run a small implant.
Turns out, our bodily fluids contain a lot of energy for the taking. Enzymatic biofuel cells (EFC) are already undergoing animal tests for their ability to metabolize energy-rich molecules for electricity generation. Via oxidation, the enzymes in the EFC force electrons into an electrode, which generates a small current. Plasma (the colorless, liquid component of blood) contains dissolved glucose, which many EFCs have been designed to use. Sweat contains lactate, which can power EFCs, but most of us don't sweat continuously. So, that avenue may fizzle. Tears, however, contain a cocktail of energy-rich molecules—and our eyes produce them constantly in minute amounts. Already, bioengineers at the University of Utah have developed an electrode-containing contact lens that "maintained a power output of over one microwatt for three hours" when exposed to a synthetic tear solution.
Kinetic Energy and Piezoelectric Materials
In addition to molecular energy contained in our fluids, we produce kinetic energy whenever we move. Even our internal organs produce enough motion that a group of Chinese and American researchers succeeded in harvesting a microwatt of power from "the beating hearts, lungs, and diaphragms of (sedated) cows and sheep." The mechanism used? Each slumbering beast had an ultra-thin piezoelectric material attached to the respective organ of study.
Piezoelectric (the Greek piezo means to squeeze or press) materials generate a charge when placed under stress. How? Such materials have an atomic lattice structure. Normally, the symmetrical structure of the lattice results in an energetic equilibrium. No difference in potential, thus, no electricity generation. However, when a force deforms the lattice—piezo—a difference in electrical potential emerges—an electrical charge.
In another experiment, researchers lined shoes with an elastomer-based piezoelectric fabric. They powered thirty LED lights from the energy generated by walking. The same group charged a lithium-ion battery by moving a shirt that had piezoelectric fabric applied to it.
Though their application to biomedicine is relatively new, piezoelectric materials have long been used in a number of industrial applications, from optics to motors to robotics. Typically, they reside in a fixed location in crystal form, creating an electrical charge when mechanically stimulated. In addition to their industrial utility, researchers have tried to scale piezoelectric technologies for macro power generation. These efforts have not been successful. Piezoelectric phenomena, it seems, operate under energetic size constraints, which is fine for developers of bioelectronic implants.
Theoretically, then, nothing prevents us from powering our bioelectric implants with hi-tech fabric attached to our organs or piezoelectric clothing that somehow—and here lies the big knowledge gap—transmits electricity to the inside of our bodies. One way or another, piezoelectric materials will almost certainly have prominent applications in the coming bioelectronic revolution.
A Matter of Time
With such attention and resources behind bioelectronic implant development, we can expect even more breakthroughs. Each of the barriers to functional bioelectronic devices—power sources; nerve-implant interfaces; durable, non-degrading materials; sufficiently sophisticated computers within the implants to read and process nerve signals and adjust output accordingly—can be individually solved, given enough research. While these challenges are significant, in the scope of human invention, they're far from insurmountable.
If, in the future, we've got these small medical devices inside of each of us, assisting our bodies with control of function, does that make us cyborgs? Invisible cyborgs, perhaps, because we'd lack the external post-apocalyptic cyborg accouterments of pop film. Does it really matter? Probably not, but it is fun to consider. Philosophical questions abound such as, for example, if we systematically add one piece of electromechanical equipment after another to modify our bodies, at what point do we stop being human and become mechanical? Or are we still human, even after every component of us has been replaced? Fascinating thought experiments. For now, however, if tiny medical devices can signal our nerves and help us reverse neurological disorders, we've accomplished a very good thing.
Jacob Andra is senior industry analyst for U.S. Translation Company, a localization firm that helps life sciences firms localize their products for overseas markets. He can be found on LinkedIn and Twitter.
Note: This article has been revised from a U.S. Translation Company blog post.