Ranica Arrowsmith, Associate Editor08.30.13
Researchers at Missouri University of Science and Technology have prototyped a glass orthopedic implant material, going by the moniker silicate 13-93, that is strong enough for load-bearing bone fractures. This is the first time any type of glass has demonstrated this kind of strength. The material also acts as a bone scaffold, adhering to cortical bone and growing new bone at the fracture site. Cortical bone, or compact bone, is the hard part of human bone tissue, and it makes up about 80 percent of the weight of a human skeleton.
Leading the research is Mohamed N. Rahaman, Ph.D., a professor in in the university’s department of materials science and engineering, director of the Center for Bone and Tissue Repair and Regeneration, and adjunct professor in the department of orthopedic surgery at University of Missouri-Columbia. The professor started his career in what could be called “technical ceramics,” ceramics used in engines and electronic components. About 10 to 15 years ago, Rahaman decided to get into biomaterials, an area where the potential for future innovation is great due to global aging populations.
One of the big successes of 13-93 is the fact that the researchers found a way to make glass that is potentially strong enough to bear a fully-grown human’s weight.
“Most of the biomaterials that have been used to repair bone in the past, including bioactive glasses, have strengths that are not up to standard,” Rahaman explained to Medical Product Outsourcing. “They are far weaker than that of cortical bone.”

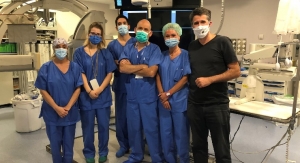
Dr. Mohamed N. (Len) Rahaman, director of the Center for Biomedical Science and Engineering, in the lab where the glass scaffold implants are fabricated. Photo courtesy of Missouri University of Science and Technology.
Two key colleagues worked with Rahaman to develop an implant material that could have real-life applications: Sonny Bal, M.D., J.D., MBA, an orthopedic surgeon and associate professor of orthopedic surgery at the University of Missouri-Columbia School of Medicine, provided insight into clinical relevance of the implant; and bone biologist Lynda Bonewald, vice chancellor for translational and clinical research at the University of Missouri-Kansas City and director of the bone biology research program the university’s School of Dentistry, evaluated the quality of the new tissue 13-93 would create after implantation.
After some years of research, Rahaman said, it became clear that the methods the team was using to build this glass would not allow for the strength and mechanical properties of cortical bone. But two years ago, the university gained some funding that allowed the team to acquire a rapid prototyping machine—better known as a 3-D printer. This particular technology, called “robocasting,” was developed in the late 1990s by Sandia National Laboratories in Albuquerque, N.M. A 1999 technology review conducted by Massachusetts Institute of Technology highlighted the prototyping machine’s particular ability to create ceramic parts denser than those made by other prototyping methods.
“We take the bioactive glass, we grind it into fine particles, then we mix it with some polymeric type additives to form a paste—somewhat like the consistency of toothpaste—and then it will be printed in the desire architecture by the robocasting machine,” said Rahaman. “The machine will be driven by a computer program for that architecture. Then, the paste will be extruded in the form of filaments in the desired architecture. The construct will have to be firmly treated to set the filaments into dense glass.”
Once implanted, 13-93 reacts with body fluids to convert to hydroxyapatite, a calcium bone mineral. After time, it remodels to living bone. So, as Rahaman said, it does not need to be removed like some metal implants would, and it does not degrade completely as a biodegradable polymer would.
“It’s like window glass, but it has a different composition,” said Rahaman. “Take window glass: it’s very durable, rain falls on it, and it doesn’t degrade. But if you change the composition—if you use a little less silica, and you modify other oxides—you can make it reactive, and that’s what this bioactive glass is. It actually converts, degrades and release ions like calcium ions that react with the phosphate ions of the body fluids, converting to calcium phosphate hydroxyapatite and then remodeling in situ.”

Missouri Science and Technology researchers are using small, porous glass scaffolds like these to regenerate bone.
Missouri University of Science and Technology has submitted a patent disclosure and applied for a provisional patent for the technology. The university hopes to submit a full patent application by January next year.
The glass has already been implanted in rat femurs in a pilot study that lasted 12 weeks. The researchers created a loaded bone defect by removing a portion of the rats’ femur and replacing it with the glass implant material, affixed with wire. Rahaman called the results “encouraging.” There is still quite a journey to in-man trials—the next steps will be implanting in progressively larger animals such as rabbits, dogs and goats.
In the future, Rahaman plans to experiment with modified glass scaffolds to see how well they enhance certain attributes within bone. For instance, doping the glass with copper should promote the growth of blood vessels or capillaries within the new bone, while doping the glass with silver will give it antibacterial properties.
The research team’s article, “Enhanced bone regeneration in rat calvarial defects implanted with surface-modified and BMP-loaded bioactive glass (13-93) scaffolds,” was published in the July issue of Acta Biomaterialia (Volume 9, Issue 7) and is available online.
Leading the research is Mohamed N. Rahaman, Ph.D., a professor in in the university’s department of materials science and engineering, director of the Center for Bone and Tissue Repair and Regeneration, and adjunct professor in the department of orthopedic surgery at University of Missouri-Columbia. The professor started his career in what could be called “technical ceramics,” ceramics used in engines and electronic components. About 10 to 15 years ago, Rahaman decided to get into biomaterials, an area where the potential for future innovation is great due to global aging populations.
One of the big successes of 13-93 is the fact that the researchers found a way to make glass that is potentially strong enough to bear a fully-grown human’s weight.
“Most of the biomaterials that have been used to repair bone in the past, including bioactive glasses, have strengths that are not up to standard,” Rahaman explained to Medical Product Outsourcing. “They are far weaker than that of cortical bone.”
Dr. Mohamed N. (Len) Rahaman, director of the Center for Biomedical Science and Engineering, in the lab where the glass scaffold implants are fabricated. Photo courtesy of Missouri University of Science and Technology.
After some years of research, Rahaman said, it became clear that the methods the team was using to build this glass would not allow for the strength and mechanical properties of cortical bone. But two years ago, the university gained some funding that allowed the team to acquire a rapid prototyping machine—better known as a 3-D printer. This particular technology, called “robocasting,” was developed in the late 1990s by Sandia National Laboratories in Albuquerque, N.M. A 1999 technology review conducted by Massachusetts Institute of Technology highlighted the prototyping machine’s particular ability to create ceramic parts denser than those made by other prototyping methods.
“We take the bioactive glass, we grind it into fine particles, then we mix it with some polymeric type additives to form a paste—somewhat like the consistency of toothpaste—and then it will be printed in the desire architecture by the robocasting machine,” said Rahaman. “The machine will be driven by a computer program for that architecture. Then, the paste will be extruded in the form of filaments in the desired architecture. The construct will have to be firmly treated to set the filaments into dense glass.”
Once implanted, 13-93 reacts with body fluids to convert to hydroxyapatite, a calcium bone mineral. After time, it remodels to living bone. So, as Rahaman said, it does not need to be removed like some metal implants would, and it does not degrade completely as a biodegradable polymer would.
“It’s like window glass, but it has a different composition,” said Rahaman. “Take window glass: it’s very durable, rain falls on it, and it doesn’t degrade. But if you change the composition—if you use a little less silica, and you modify other oxides—you can make it reactive, and that’s what this bioactive glass is. It actually converts, degrades and release ions like calcium ions that react with the phosphate ions of the body fluids, converting to calcium phosphate hydroxyapatite and then remodeling in situ.”
Missouri Science and Technology researchers are using small, porous glass scaffolds like these to regenerate bone.
The glass has already been implanted in rat femurs in a pilot study that lasted 12 weeks. The researchers created a loaded bone defect by removing a portion of the rats’ femur and replacing it with the glass implant material, affixed with wire. Rahaman called the results “encouraging.” There is still quite a journey to in-man trials—the next steps will be implanting in progressively larger animals such as rabbits, dogs and goats.
In the future, Rahaman plans to experiment with modified glass scaffolds to see how well they enhance certain attributes within bone. For instance, doping the glass with copper should promote the growth of blood vessels or capillaries within the new bone, while doping the glass with silver will give it antibacterial properties.
The research team’s article, “Enhanced bone regeneration in rat calvarial defects implanted with surface-modified and BMP-loaded bioactive glass (13-93) scaffolds,” was published in the July issue of Acta Biomaterialia (Volume 9, Issue 7) and is available online.