01.17.12
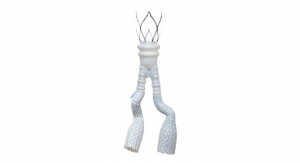
Medtronic Inc. has the green light in Europe for its Endurant II stent graft system for abdominal aortic aneurysm (AAA). With its recent CE mark the company, is ready for the international rollout of the device, which, according to the company expands options for physicians outside the United States to treat patients with AAA through a minimally invasive technique called endovascular aortic repair.
AAA is a weakening or bulge in the segment of the aorta, the body’s main artery that crosses through the abdomen. AAA often is called a “silent killer” because it rarely causes symptoms until it ruptures, which often causes death.
Developed in collaboration with more than 250 physicians from around the world, the new system encompasses the clinical performance of the first-generation Endurant stent graft platform (approved in the United States in December 2010), while adding design features that enhance the device’s ease of use, company officials noted.
According to a press release, the Endurant II AAA stent graft system adds three enhancements to the Endurant line:
• Beginning at the point of access, the new lower-profile delivery system –– with 35 percent extended hydrophilic coating for enhanced access to challenging anatomies –– allows the 28 mm-diameter bifurcated segment (the most commonly used size) to fit inside an 18 French outer diameter catheter (down from 20 French with the original device);
• Two added contralateral limb lengths (156 mm and 199 mm) enable more configuration options requiring fewer total pieces; and
• The radiopacity of the distal end of the bifurcated segment’s contralateral gate has been improved to enhance visibility and aid with limb insertion, placement and deployment.
“The Endurant II AAA Stent Graft System will confer considerable confidence to vascular surgeons who use EVAR to treat even the most complex AAAs,” said Hence Verhagen, professor and chief of vascular surgery at the Erasmus Medical Center in Rotterdam, the Netherlands. “Building on the exceptional clinical outcomes of the original system, which has significantly increased the applicability of EVAR, Endurant II offers an even better user experience which will benefit even more patients whose AAAs are detected before rupturing.”
Verhagen, who led the European clinical trial of the original Endurant Stent Graft that contributed to that device’s approval, was the first physician to successfully use the new system since it received the CE mark.
In countries where the Endurant II has CE mark, the device is indicated for the endovascular treatment of abdominal aortic aneurysms in patients with a proximal neck ≥ 10 mm in length with ≤ 60-degree infrarenal and ≤ 45-degree suprarenal angulation and in patients with a proximal aortic neck ≥ 15 mm in length with ≤ 75-degree infrarenal and ≤ 60-degree suprarenal angulation.
Officials at Minneapolis, Minn.-based Medtronic expect Endurant II to receive premarket approval in the United States this year.
AAA is a weakening or bulge in the segment of the aorta, the body’s main artery that crosses through the abdomen. AAA often is called a “silent killer” because it rarely causes symptoms until it ruptures, which often causes death.
Developed in collaboration with more than 250 physicians from around the world, the new system encompasses the clinical performance of the first-generation Endurant stent graft platform (approved in the United States in December 2010), while adding design features that enhance the device’s ease of use, company officials noted.
According to a press release, the Endurant II AAA stent graft system adds three enhancements to the Endurant line:
• Beginning at the point of access, the new lower-profile delivery system –– with 35 percent extended hydrophilic coating for enhanced access to challenging anatomies –– allows the 28 mm-diameter bifurcated segment (the most commonly used size) to fit inside an 18 French outer diameter catheter (down from 20 French with the original device);
• Two added contralateral limb lengths (156 mm and 199 mm) enable more configuration options requiring fewer total pieces; and
• The radiopacity of the distal end of the bifurcated segment’s contralateral gate has been improved to enhance visibility and aid with limb insertion, placement and deployment.
“The Endurant II AAA Stent Graft System will confer considerable confidence to vascular surgeons who use EVAR to treat even the most complex AAAs,” said Hence Verhagen, professor and chief of vascular surgery at the Erasmus Medical Center in Rotterdam, the Netherlands. “Building on the exceptional clinical outcomes of the original system, which has significantly increased the applicability of EVAR, Endurant II offers an even better user experience which will benefit even more patients whose AAAs are detected before rupturing.”
Verhagen, who led the European clinical trial of the original Endurant Stent Graft that contributed to that device’s approval, was the first physician to successfully use the new system since it received the CE mark.
In countries where the Endurant II has CE mark, the device is indicated for the endovascular treatment of abdominal aortic aneurysms in patients with a proximal neck ≥ 10 mm in length with ≤ 60-degree infrarenal and ≤ 45-degree suprarenal angulation and in patients with a proximal aortic neck ≥ 15 mm in length with ≤ 75-degree infrarenal and ≤ 60-degree suprarenal angulation.
Officials at Minneapolis, Minn.-based Medtronic expect Endurant II to receive premarket approval in the United States this year.