08.12.10
The Tempe, Ariz.-based provider of mechanical engineering services has purchased the building in which it has resided for the last eight years. Executives said the 48,000-square-foot facility in the Arizona State University Research Park will enable its medical device group (aptly named PADT Medical) to incubate a greater number of medical device startups.
“The ASU Research Park is a superb location for us,’’ said Ward Rand, PADT president. “The purchase lowers our operating costs and enables more freedom to offer incubation space to local startups.’’
PADT Medical provides startup device firms with research and development services to help accelerate product design, build clinical-use prototypes, perform validation testing and meet regulatory compliance. PADT formed the medical device group in 2006; the group has been compliant with U.S. Food and Drug Administration standards since 2007.
Founded in 1994 by aerospace engineers, PADT has helped nearly three dozen companies develop products and bring them to market. Customers include C.R. Bard Inc., Medtronic Inc., St. Jude Medical Inc., W.L. Gore & Associates Inc., Orthologic Corp., Kinetic Concepts Inc., GE Medical Systems, Accutron Inc., and C.R. Technologies Inc.
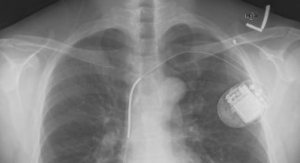
The number of medical devices PADT has helped bring to market is almost as long as the list of companies it has partnered with to offer its services. According to its website, PADT has contributed to the development of a heart valve stent, a blood filter device, an implantable pacemaker, a pediatric dentist drill, a bone implant and bone screw, a bone paste delivery device, a heated catheter and a heart valve implant, among others.
“The ASU Research Park is a superb location for us,’’ said Ward Rand, PADT president. “The purchase lowers our operating costs and enables more freedom to offer incubation space to local startups.’’
PADT Medical provides startup device firms with research and development services to help accelerate product design, build clinical-use prototypes, perform validation testing and meet regulatory compliance. PADT formed the medical device group in 2006; the group has been compliant with U.S. Food and Drug Administration standards since 2007.
Founded in 1994 by aerospace engineers, PADT has helped nearly three dozen companies develop products and bring them to market. Customers include C.R. Bard Inc., Medtronic Inc., St. Jude Medical Inc., W.L. Gore & Associates Inc., Orthologic Corp., Kinetic Concepts Inc., GE Medical Systems, Accutron Inc., and C.R. Technologies Inc.
The number of medical devices PADT has helped bring to market is almost as long as the list of companies it has partnered with to offer its services. According to its website, PADT has contributed to the development of a heart valve stent, a blood filter device, an implantable pacemaker, a pediatric dentist drill, a bone implant and bone screw, a bone paste delivery device, a heated catheter and a heart valve implant, among others.