12.21.05
Toms River, NJ-based OEM Concepts' manufacturing facility can produce up to three kilograms each year. Its capacity exemplifies how contract bioprocessors' economies of scale are paving the way for many diagnostics companies to outsource portions of their manufacturing. It's also why many bioprocessors are finding being a niche player in the diagnostics industry isn't a bad thing.
As the industry continues to look for cost-cutting measures and access to new technology, the services of bioprocessors seem to make sense. With their ability to turn out large quantities of a specific antibody, protein or control serum at a low cost, clients find it more economical to outsource than invest in costly capital equipment and personnel. Furthermore, contract manufacturers are able to help IVD companies scale up more quickly.
While it may seem that outsourcing is accelerating in the industry, the opposite might be true at some companies, especially large, quality-sensitive diagnostics marketers. That's because quality concerns are overshadowing the economic benefits of outsourcing at some firms, and control, not cost, is more valued.
One IVD Maker's View
"Outsourcing makes sense if you are trying to control costs," said Kent Kost, vice president of operations and site manager at Indianapolis-based Roche Diagnostics, the world's largest IVD manufacturer. However, he said, the diagnostic industry these days is more concerned about quality, which in part is driven by customer requirements. Part of the company's efforts to ratchet up quality is to shift some manufacturing-particularly products made by problem-plagued vendors-to in-house facilities. He said Roche will continue to work with trusted vendors, but bad experiences have led it to sever troublesome relationships.
"Those are the very [vendors] we look at phasing out," he said.
Kost's comments reflect what some regard as a dilemma for many bioprocessing firms. A small market that one provider estimated at $25 million (excluding antibody production), the outsourced bioprocessing sector is comprised of many small niche-focused firms catering to similarly sized customers. Investing in costly ISO certification initiatives or implementing quality programs equivalent to that of their biggest customers is not feasible.
Still, they offer valuable services that many diagnostics manufacturers are unable to provide for themselves economically. And, they understand adhering to customers' quality standards is a mandate for longevity in the business.
"The most important criteria is compliance to quality standards. I don't think there has been any change to that," said Bill Morrison, vice president of sales and marketing at Newark, DE-based Strategic Biosolutions, an antibody supplier.
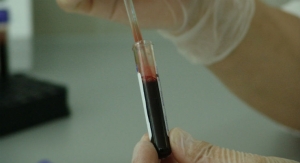
| Economies of scale help contract bioprocessors provide products to customers more cost effectively and continue to be a driver for the outsourcing trends. Above, a production personnel prepares monoclonal antibody cell cultures under a laminar flow hood. (Photo courtesy of OEM Concepts.) |
Satisfying those requisites also means bioprocessing firms must provide responsive customer service. Being able to quickly address production issues, answering customer questions and delivering products that conform to specs all help earn the trust of IVD manufacturers. However, failure to meet any of these expectations can influence a particular company's view on outsourcing, said Bob Minarchi, president of OEM Concepts.
"This is something that waxes and wanes over time. It is dictated by successes and failures," he said of differing views on outsourcing. While Minarchi said he felt the trend to outsource "is still in a positive direction," he added individual outsourcing strategies can be influenced by good and bad vendors. That's why diagnostic companies have to be careful in their selection of outsourcing partners.
For instance, he noted that some large companies outsource their excess capacity and view bioprocessing as a secondary revenue source. He emphasized that companies whose sole business is to provide for external customers tend to be more attentive than firms whose main revenue source comes from services other than bioprocessing.
As bioprocessors try to meet their customers' quality expectations, they're finding that it hasn't been easy, in part because it can be a nebulous concept, defined differently by each customer. Early on, ISO 9001 registration was thought to be a requirement for doing business, yet many companies successfully manufacturing for diagnostics companies these days don't have certification. On the other hand, there are IVD companies that don't outsource because of quality concerns even though some vendors are engaged in cGMP and ISO. In the end, quality may simply serve as a proxy for control.
Still, QA/QC have become more regimented over the years with the adoption of formal programs and audits. Through such efforts, OEMs at least have documented mechanisms for ensuring quality products from their vendors.
The demand for higher-quality outsourced products is being driven from all sides, including end users, diagnostics manufacturers and regulators worldwide. Kevin Jones, vice president of sales and marketing at Carlsbad, CA-based Aalto Scientific, a supplier of human proteins and control sera, said toughened regulatory requirements for manufacturers in Europe and the U.S. in part help the industry ensure products meet quality standards.
"Government regulations in the U.S. and the rest of the world have gotten so stringent that quality has to be built into the products at this point," he said, adding that Aalto is ISO 9001 certified.
Control is certainly a key issue for IVD companies. Many consider manufacturing part of their core competencies and are reluctant to hand over those responsibilities to a third party, no matter how efficiently it operates. Companies with production capabilities tend to perform bioprocessing functions internally for mainstream products and leave the manufacturing of specialized products to outside experts. As a result, technology-rich bioprocessors stand to win more contracts as the industry becomes more selective in its outsourcing strategy.
Outsourcing Specialties
To be sure, Kost said large IVD companies such as Roche will look to keep in-house the manufacture of "commodity" types of products for quality reasons. However, when it comes to products with highly protected intellectual property, IVD manufacturers will have no other choice but outsource. "To the extent that somehow we would have restricted access [to the technology], that would be an OEM opportunity," he added.
To develop those opportunities, bioprocessors are focusing on niche areas. Gary Goodrich, president of Bioprocessing, Inc., a Scarborough, ME-based raw materials supplier, said he believes that only tightly focused bioprocessors will be successful in the industry. Unlike the medical device sector, IVD manufacturers don't want one-stop shops; they are looking for specialists.
"We've had good success where once we get their attention and we have a service they need, then in an audit situation they see we are narrowly focused," Goodrich said.
Goodrich's company specializes in oncology tumor markers, which he said is experiencing tremendous growth on the research side, but has yet to reach high volumes on the commercial side. Still, he said, diagnostics companies may see rapid growth in the future if they can partner with up-and-coming cancer therapy developers. Companion diagnostics treatments, he added, could yield a promising market, especially if a particular drug therapy is an early winner.
For bioprocessing firms, demand for products such as recombinant proteins could potentially explode, offering suppliers with the right expertise tremendous growth opportunities.
Growth in other technological sectors is also spurring a rise in outsourcing. In molecular diagnostics, the demand for control assays is accelerating as new tests are continually introduced to the market. One emerging challenge for a number of manufacturers is developing controls and standards at low copy numbers.
Some bioprocessing firms such as Ambion Diagnostics have stepped into the fray by developing new technology to standardize the process, said Peter Donahue, director of business development at the Austin, TX-based company. A reagent maker and specialist in molecular diagnostics, the company often develops QC assays for the materials it sells, said Donahue. He added that diagnostics companies are increasingly turning to Ambion for this type of expertise simply because they don't have the resources.
"I think it's time constraints; they're overworked," he said. "They don't have time to think about [these issues]."
More research has also helped bioprocessing firms grow commercial business. Some companies say as customers develop successful products, their needs increase exponentially when they scale up to commercial production. For the contract manufacturer, that means much higher revenues as well.
Bioprocessors say the sooner they begin work with customers on new products, the more efficiently they can manufacture. Strategic Biosolution's Morrison said his company prefers to hold discussions before cell banks are even established for the hybrid line. More and more, customers are willing to engage their partners early on. "I perceive an increasing realization that if antibody production is going to be outsourced, it's important to involve that partner as soon as possible in the product development process.," he added.
Even though the outsourcing of bioprocessing services continues to grow, contract firms must still address quality concerns diagnostics manufacturers harbor. To allay those fears, they will need to provide documented quality systems, offer responsive customer service and remain focused in their area of expertise.