Michael Barbella, Managing Editor04.11.24
Procyrion Inc. is $57.7 million richer these days, having closed a Series E funding round.
This most recent financing included the conversion of $10 million in interim financing. The funds will help support the DRAIN-HF pivotal Investigational Device Exemption (IDE) trial evaluating the company's Aortix percutaneous mechanical circulatory support (pMCS) device in patients with acute decompensated heart failure (ADHF) who remain congested despite standard medical therapy (cardiorenal syndrome or CRS). In addition, the money will finance internal programs to improve product manufacturability and prepare for commercialization. The round was led by Fannin Partners in partnership with new and existing family/multi-family office investors. The Series E financing also included significant participation from returning investors including Bluebird Ventures and an undisclosed strategic investor.
"Approximately 25% of the millions of patients admitted to the hospital with ADHF are unable to be successfully treated with standard of care therapies, yet there is a lack of effective treatment options available, leading to very poor outcomes. Aortix therapy is uniquely suited for treating CRS patients and this latest round of investment will enable the company to make significant progress toward commercialization of our technology," Procyrion President/CEO Eric S. Fain, M.D., said. "We thank our investors for recognizing the engineering and clinical achievements to date, as well as the potential of Aortix to be an advancement to break the vicious CRS cycle and improve the outcomes in these most challenging-to-manage heart failure patients who currently have no proven therapeutic options."
Patients with CRS—characterized by resistance to standard of care intravenous diuretic therapy1—experience a vicious cycle: The underlying heart failure leads to reduced blood flow to the kidneys, causing the kidneys to become less effective at bodily fluid removal, which further stresses the heart. Effective treatment options for CRS are lacking, as evidenced by up to a 25% rate for heart failure rehospitalization or mortality at 30 days2,3 for those patients who remained clinically congested after 96 hours of standard of care medical therapy and were discharged from the hospital.1
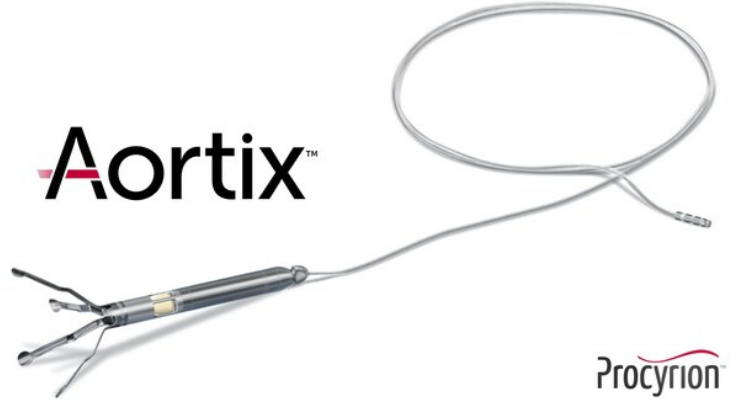
Aortix is a catheter-deployed pump that is placed in the descending thoracic aorta and designed to directly increase perfusion to the kidneys while unloading the heart and improving cardiac performance. Its design harnesses fluid entrainment to pump blood without the need for a valve, delivering a therapeutic benefit through a physiologically natural mechanism of action. In the Aortix CRS Pilot Study, published in the Journal of the American College of Cardiology: Heart Failure, patients demonstrated rapid decongestion with substantial removal of excess fluid and improved hemodynamics, as well as significant improvements in kidney function, cardiac function, and patient-reported assessment of shortness of breath sustained out to 30 days after treatment with the Aortix pump.
"Approximately 500,000 patients in the U.S. who are admitted to the hospital with cardiorenal syndrome are unable to be successfully treated with current available therapies and suffer high rates of mortality and heart failure rehospitalization," Fannin Partners Chairman/Founder Leo Linbeck III, a Procyrion board member. "We are pleased to lead this round as we see enormous commercial potential for Aortix and look forward to seeing the results of the DRAIN-HF study with the goal of demonstrating improved patient outcomes, which should also reduce the burden on the overall healthcare system."
Procyrion Inc. is a privately held medical device company providing cardiac and renal impairment solutions. Its Aortix percutaneous mechanical circulatory support device is a catheter-deployed pump technology that harnesses fluid entrainment to pump blood to address multiple conditions. Aortix is limited by law to investigational use only and is not approved for sale in any geography.
Fannin is a life science product development group, with a dozen programs/platforms at different stages of development. Fannin advances its pipeline both internally and through Fannin-founded entities. In the last decade, Fannin programs have sourced approximately $225 million of funding, with about $75 million from grants and $150 million from private investors. Fannin's talent development program has also grown to become one of the largest of its kind, with more than 320 alumni active in pharma/biotech, medical device/medtech, and venture capital.
References
1 Cooper et al. Clin Cardiol. 2023 Jul 18. DOI: 10.1002/clc.24080
2 Bart et al., N Engl J Med 2012; DOI: 10.1056/NEJMoa1210357
3 Wattad et al., Am Journal of Cardiol 2015; DOI: 10.1016/j.amjcard.2015.01.019
This most recent financing included the conversion of $10 million in interim financing. The funds will help support the DRAIN-HF pivotal Investigational Device Exemption (IDE) trial evaluating the company's Aortix percutaneous mechanical circulatory support (pMCS) device in patients with acute decompensated heart failure (ADHF) who remain congested despite standard medical therapy (cardiorenal syndrome or CRS). In addition, the money will finance internal programs to improve product manufacturability and prepare for commercialization. The round was led by Fannin Partners in partnership with new and existing family/multi-family office investors. The Series E financing also included significant participation from returning investors including Bluebird Ventures and an undisclosed strategic investor.
"Approximately 25% of the millions of patients admitted to the hospital with ADHF are unable to be successfully treated with standard of care therapies, yet there is a lack of effective treatment options available, leading to very poor outcomes. Aortix therapy is uniquely suited for treating CRS patients and this latest round of investment will enable the company to make significant progress toward commercialization of our technology," Procyrion President/CEO Eric S. Fain, M.D., said. "We thank our investors for recognizing the engineering and clinical achievements to date, as well as the potential of Aortix to be an advancement to break the vicious CRS cycle and improve the outcomes in these most challenging-to-manage heart failure patients who currently have no proven therapeutic options."
Patients with CRS—characterized by resistance to standard of care intravenous diuretic therapy1—experience a vicious cycle: The underlying heart failure leads to reduced blood flow to the kidneys, causing the kidneys to become less effective at bodily fluid removal, which further stresses the heart. Effective treatment options for CRS are lacking, as evidenced by up to a 25% rate for heart failure rehospitalization or mortality at 30 days2,3 for those patients who remained clinically congested after 96 hours of standard of care medical therapy and were discharged from the hospital.1
Aortix is a catheter-deployed pump that is placed in the descending thoracic aorta and designed to directly increase perfusion to the kidneys while unloading the heart and improving cardiac performance. Its design harnesses fluid entrainment to pump blood without the need for a valve, delivering a therapeutic benefit through a physiologically natural mechanism of action. In the Aortix CRS Pilot Study, published in the Journal of the American College of Cardiology: Heart Failure, patients demonstrated rapid decongestion with substantial removal of excess fluid and improved hemodynamics, as well as significant improvements in kidney function, cardiac function, and patient-reported assessment of shortness of breath sustained out to 30 days after treatment with the Aortix pump.
"Approximately 500,000 patients in the U.S. who are admitted to the hospital with cardiorenal syndrome are unable to be successfully treated with current available therapies and suffer high rates of mortality and heart failure rehospitalization," Fannin Partners Chairman/Founder Leo Linbeck III, a Procyrion board member. "We are pleased to lead this round as we see enormous commercial potential for Aortix and look forward to seeing the results of the DRAIN-HF study with the goal of demonstrating improved patient outcomes, which should also reduce the burden on the overall healthcare system."
Procyrion Inc. is a privately held medical device company providing cardiac and renal impairment solutions. Its Aortix percutaneous mechanical circulatory support device is a catheter-deployed pump technology that harnesses fluid entrainment to pump blood to address multiple conditions. Aortix is limited by law to investigational use only and is not approved for sale in any geography.
Fannin is a life science product development group, with a dozen programs/platforms at different stages of development. Fannin advances its pipeline both internally and through Fannin-founded entities. In the last decade, Fannin programs have sourced approximately $225 million of funding, with about $75 million from grants and $150 million from private investors. Fannin's talent development program has also grown to become one of the largest of its kind, with more than 320 alumni active in pharma/biotech, medical device/medtech, and venture capital.
References
1 Cooper et al. Clin Cardiol. 2023 Jul 18. DOI: 10.1002/clc.24080
2 Bart et al., N Engl J Med 2012; DOI: 10.1056/NEJMoa1210357
3 Wattad et al., Am Journal of Cardiol 2015; DOI: 10.1016/j.amjcard.2015.01.019