Sam Brusco, Associate Editor03.27.24
SyntheticMR has received U.S. Food and Drug Administration (FDA) 510(k) clearance for its next-gen imaging solutions with isotropic resolution, SyMRI 3D.
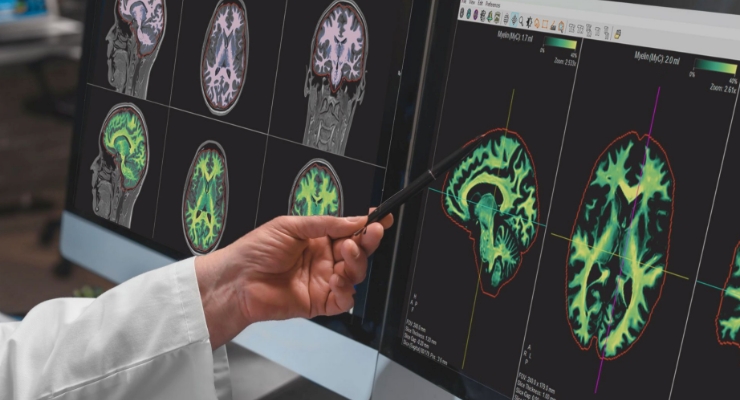
SyMRI 3D allows precise, volumetric brain region estimation, which is commonly referred to as parcellation. This helps clinicians gain more insights into brain structure and function. SyMRI 3D’s resolution also facilitates comprehensive lesion analysis, according to the company.
The company said the milestone of FDA clearance marks a significant advancement for quantitative MRI technology with unprecedented resolution and accuracy in brain imaging.
"We are thrilled to receive 510(k) clearance for SyMRI 3D," said Ulrik Harrysson, CEO at SyntheticMR. "SyMRI 3D represents the next generation of quantitative MRI, revolutionizing the landscape of medical diagnostics and offering new possibilities for diagnosis and treatment."
SyMRI 3D can open new possibilities for precise diagnosis, treatment planning, and monitoring to improve outcomes.
"Receiving 510(k) clearance for SyMRI 3D allows us to empower physicians to make more precise and informed decisions in diagnosis and treatment planning through quantitative imaging," said Jared Dixon, president of SyntheticMR U.S Inc.
SyMRI is available in different packages. SyMRI NEURO delivers multiple contrast images, tissue segmentations and quantitative data on the brain. SyMRI MSK provides multiple contrast images and quantitative data for knee and spine anatomies.
SyMRI 3D allows precise, volumetric brain region estimation, which is commonly referred to as parcellation. This helps clinicians gain more insights into brain structure and function. SyMRI 3D’s resolution also facilitates comprehensive lesion analysis, according to the company.
The company said the milestone of FDA clearance marks a significant advancement for quantitative MRI technology with unprecedented resolution and accuracy in brain imaging.
"We are thrilled to receive 510(k) clearance for SyMRI 3D," said Ulrik Harrysson, CEO at SyntheticMR. "SyMRI 3D represents the next generation of quantitative MRI, revolutionizing the landscape of medical diagnostics and offering new possibilities for diagnosis and treatment."
SyMRI 3D can open new possibilities for precise diagnosis, treatment planning, and monitoring to improve outcomes.
"Receiving 510(k) clearance for SyMRI 3D allows us to empower physicians to make more precise and informed decisions in diagnosis and treatment planning through quantitative imaging," said Jared Dixon, president of SyntheticMR U.S Inc.
SyMRI is available in different packages. SyMRI NEURO delivers multiple contrast images, tissue segmentations and quantitative data on the brain. SyMRI MSK provides multiple contrast images and quantitative data for knee and spine anatomies.