Michael Barbella, Managing Editor03.18.24
Hyperfine Inc. has officially launched the eighth generation of its AI-powered Swoop system software.
The software’s diffusion-weighted imaging (DWI) AI denoising feature received U.S. Food and Drug Administration (FDA) clearance in October 2023, and has been rolled out at select sites through a limited market release. The latest software improves Swoop system image quality and introduces valuable ease-of-use features, including a real-time aid for more precise patient loading and positioning and a streamlined image upload process.
With CE and UKCA certifications for this latest software, Hyperfine Inc. is well positioned for future international expansion, bringing the Swoop system to a broader global market.
“Our latest AI-powered software, the eighth generation of our proprietary software platform, embodies our commitment to supporting clinicians in critical decision-making,” Hyperfine Chief Operating Officer Tom Teisseyre, Ph.D., said. “Our focus on image quality with this latest software has been on the DWI sequence, which is key in stroke imaging. Since its first FDA clearance in 2020, we’ve been dedicated to continually enhancing image quality and workflow efficiencies to define best-in-class, user-centric, ultra-low field MR brain imaging.”
More than 50 exams were performed in hospitals taking part in the limited market release phase, and users commented favorably on DWI image quality. Participating sites confirmed that the new streamlined upload feature materially shortened upload completion times, and the fast-positioning check was a time-saving aid and helped improve image acquisition.
“The software update on the Swoop system has been most beneficial to our workflow. We are now able to view each series as it is being performed and can communicate with the technologist whether additional series are needed. Additionally, we can communicate with clinicians any critical findings in real-time without having to wait for the whole study to be completed,” noted Jennifer Villa Frabizzio M.D., a neuroradiologist with Radiology Group of Abington.
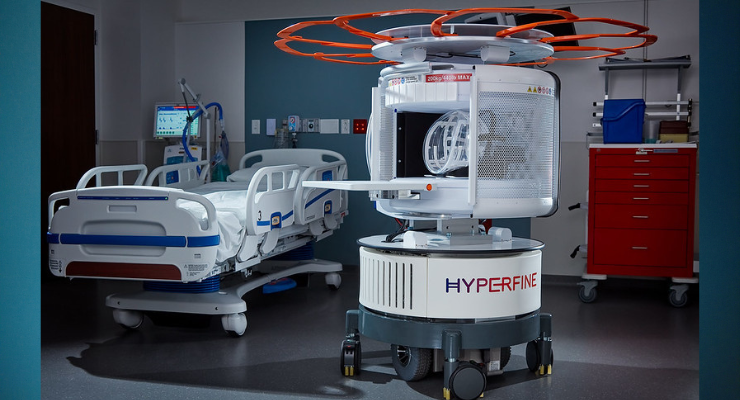
Hyperfine Inc. has redefined brain imaging with the Swoop system—the world’s first FDA-cleared, portable, ultra-low-field, magnetic resonance brain imaging system capable of providing imaging at multiple points of care. The Swoop system received initial FDA clearance in 2020. It is a portable ultra-low-field magnetic resonance brain imaging device for producing images that display the internal structure of the head where a full diagnostic examination is not clinically practical. When interpreted by a trained physician, these images provide information that can be useful in determining a diagnosis. The Swoop system has been approved for brain imaging in several countries, including Canada and Australia, has UKCA certification in the United Kingdom, CE certification in the European Union, and is also available in New Zealand.
Hyperfine aims to revolutionize patient care globally through transformational, accessible, clinically relevant diagnostic imaging and data solutions. Founded by Dr. Jonathan Rothberg in a technology-based incubator called 4Catalyzer, Hyperfine scientists, engineers, and physicists developed the Swoop system out of a passion for redefining brain imaging methodology and the ways clinicians can apply accessible diagnostic imaging to patient care.
The software’s diffusion-weighted imaging (DWI) AI denoising feature received U.S. Food and Drug Administration (FDA) clearance in October 2023, and has been rolled out at select sites through a limited market release. The latest software improves Swoop system image quality and introduces valuable ease-of-use features, including a real-time aid for more precise patient loading and positioning and a streamlined image upload process.
With CE and UKCA certifications for this latest software, Hyperfine Inc. is well positioned for future international expansion, bringing the Swoop system to a broader global market.
“Our latest AI-powered software, the eighth generation of our proprietary software platform, embodies our commitment to supporting clinicians in critical decision-making,” Hyperfine Chief Operating Officer Tom Teisseyre, Ph.D., said. “Our focus on image quality with this latest software has been on the DWI sequence, which is key in stroke imaging. Since its first FDA clearance in 2020, we’ve been dedicated to continually enhancing image quality and workflow efficiencies to define best-in-class, user-centric, ultra-low field MR brain imaging.”
More than 50 exams were performed in hospitals taking part in the limited market release phase, and users commented favorably on DWI image quality. Participating sites confirmed that the new streamlined upload feature materially shortened upload completion times, and the fast-positioning check was a time-saving aid and helped improve image acquisition.
“The software update on the Swoop system has been most beneficial to our workflow. We are now able to view each series as it is being performed and can communicate with the technologist whether additional series are needed. Additionally, we can communicate with clinicians any critical findings in real-time without having to wait for the whole study to be completed,” noted Jennifer Villa Frabizzio M.D., a neuroradiologist with Radiology Group of Abington.
Hyperfine Inc. has redefined brain imaging with the Swoop system—the world’s first FDA-cleared, portable, ultra-low-field, magnetic resonance brain imaging system capable of providing imaging at multiple points of care. The Swoop system received initial FDA clearance in 2020. It is a portable ultra-low-field magnetic resonance brain imaging device for producing images that display the internal structure of the head where a full diagnostic examination is not clinically practical. When interpreted by a trained physician, these images provide information that can be useful in determining a diagnosis. The Swoop system has been approved for brain imaging in several countries, including Canada and Australia, has UKCA certification in the United Kingdom, CE certification in the European Union, and is also available in New Zealand.
Hyperfine aims to revolutionize patient care globally through transformational, accessible, clinically relevant diagnostic imaging and data solutions. Founded by Dr. Jonathan Rothberg in a technology-based incubator called 4Catalyzer, Hyperfine scientists, engineers, and physicists developed the Swoop system out of a passion for redefining brain imaging methodology and the ways clinicians can apply accessible diagnostic imaging to patient care.