Sam Brusco, Associate Editor03.08.24
The U.S. Food and Drug Administration (FDA) has identified Medtronic’s January 22 recall of its Duet external drainage and monitoring system (EDMS) catheter tubing as Class I.
The Duet EDMS is used to temporarily drain cerebrospinal fluid (CSF) or for CSF sampling in patients who have surgery for open descending thoracic aortic aneurysm (open TAA) or open descending thoraco-abdominal aortic aneurysm (open TAAA). It’s also used for patients who had TAA/TAAA repair surgery and develop symptoms like paraplegia.
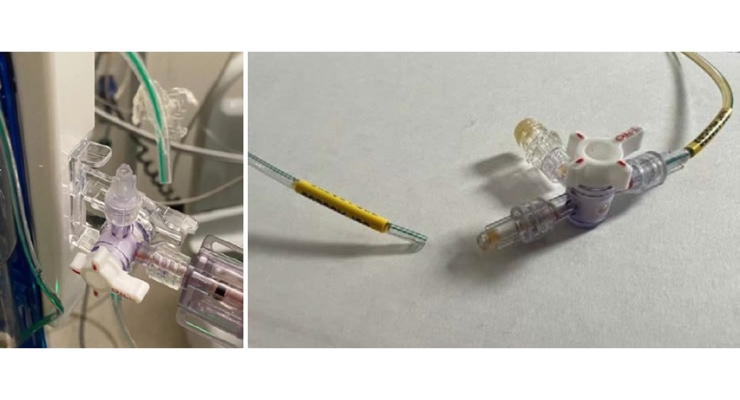
The system is comprised of a green-striped tube, stopcocks, a drip chamber, a pressure scale, needleless injection/CSF sampling sites, and a removable bag with measurements and an air vent. It works by gravity, flowing CSF from an external lumbar catheter though the tube, into the drip chamber, and into the bag.
Medtronic recalled the Duet EDMS catheter tubing because of the risk of disconnection from the patient line stopcock connectors. If this happens, risks to patients include infections, cerebrospinal fluid leakage, over drainage of cerebrospinal fluid, and abnormality of the ventricles. Uncontrolled over drainage of CSF can lead to neurological injury or death if the disconnection isn’t detected.
Medtronic has so far reported 26 injuries related to the issue, and no reports of death.
Users were advised to check all components for damage and make sure all connections are secure and leak-free. If a patient is connected to an impacted device with a leak or disconnection, an alternative device should be used with a sterile technique.
Medtronic also said any unused, impacted products should be identified and quarantined. The company advised against removing or replacing a Duet system that is working as intended.
The Duet EDMS is used to temporarily drain cerebrospinal fluid (CSF) or for CSF sampling in patients who have surgery for open descending thoracic aortic aneurysm (open TAA) or open descending thoraco-abdominal aortic aneurysm (open TAAA). It’s also used for patients who had TAA/TAAA repair surgery and develop symptoms like paraplegia.
The system is comprised of a green-striped tube, stopcocks, a drip chamber, a pressure scale, needleless injection/CSF sampling sites, and a removable bag with measurements and an air vent. It works by gravity, flowing CSF from an external lumbar catheter though the tube, into the drip chamber, and into the bag.
Medtronic recalled the Duet EDMS catheter tubing because of the risk of disconnection from the patient line stopcock connectors. If this happens, risks to patients include infections, cerebrospinal fluid leakage, over drainage of cerebrospinal fluid, and abnormality of the ventricles. Uncontrolled over drainage of CSF can lead to neurological injury or death if the disconnection isn’t detected.
Medtronic has so far reported 26 injuries related to the issue, and no reports of death.
Users were advised to check all components for damage and make sure all connections are secure and leak-free. If a patient is connected to an impacted device with a leak or disconnection, an alternative device should be used with a sterile technique.
Medtronic also said any unused, impacted products should be identified and quarantined. The company advised against removing or replacing a Duet system that is working as intended.