Sam Brusco, Associate Editor02.07.24
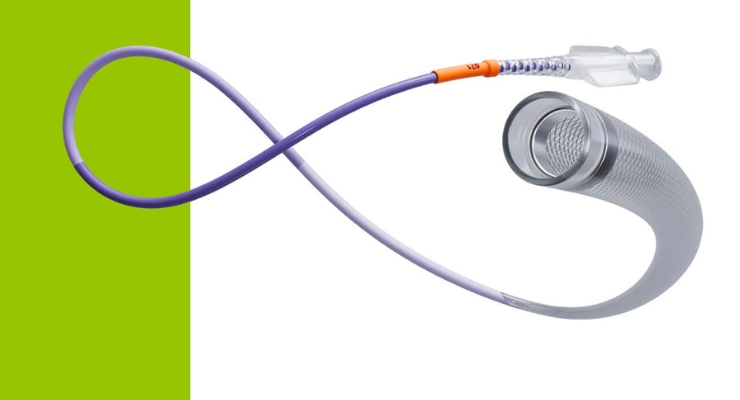
CERENOVUS, part of Johnson & Johnson MedTech, launched its CereGlide 71 intermediate catheter for revascularization of patients suffering from acute ischemic stroke.
CereGlide 71, according to the company, is optimized for effective direct aspiration and delivery of compatible stent retrievers into the neurovasculature. This includes the company’s EmboTrap III revascularization device.
The intermediate catheter features TruCourse technology to increase flexibility and improve navigation and access to clots, even in challenging anatomies. The company said TruCourse provides optimal compatibility, durable delivery, and reliable trackability during thrombectomy.
“In the rapidly evolving landscape of neuroendovascular stroke care, staying on the cutting edge is crucial for advancing the field and ensuring physicians have the tools to navigate complex patient anatomy” said Fawaz Al-Mufti, MD, Interventional Neurologist, Director of the Neuroendovascular Surgery Fellowship, Westchester Medical Center/New York Medical College. “Balancing trackability, support, and aspiration efficacy is crucial in overcoming the challenges of swift clot access during endovascular thrombectomy. As the global first user, in initial use, CEREGLIDE 71 Intermediate Catheter does just that. It impressively navigates, accesses occlusion sites, and engages with effectiveness to aspirate clots, rapidly restoring blood flow in the patient’s brain—potentially a life-saving intervention.”
CereGlide 71 will be included in the next phase of the CERENOVUS EXCELLENT registry, a real-world registry focused on study of stroke-inducing blood clot removal via mechanical thrombectomy.
“CEREGLIDE 71 Intermediate Catheter is specifically designed to glide through challenging anatomical conditions,” said Mark Dickinson, Worldwide President, CERENOVUS. “Developed through robust research and clinical insights, our team designed a catheter for physicians that addresses unmet clinical needs by providing reliable trackability, durable delivery and the versatility for both direct aspiration and stent-retriever use—even in the most challenging anatomical conditions.”
CereGlide 71, according to the company, is optimized for effective direct aspiration and delivery of compatible stent retrievers into the neurovasculature. This includes the company’s EmboTrap III revascularization device.
The intermediate catheter features TruCourse technology to increase flexibility and improve navigation and access to clots, even in challenging anatomies. The company said TruCourse provides optimal compatibility, durable delivery, and reliable trackability during thrombectomy.
“In the rapidly evolving landscape of neuroendovascular stroke care, staying on the cutting edge is crucial for advancing the field and ensuring physicians have the tools to navigate complex patient anatomy” said Fawaz Al-Mufti, MD, Interventional Neurologist, Director of the Neuroendovascular Surgery Fellowship, Westchester Medical Center/New York Medical College. “Balancing trackability, support, and aspiration efficacy is crucial in overcoming the challenges of swift clot access during endovascular thrombectomy. As the global first user, in initial use, CEREGLIDE 71 Intermediate Catheter does just that. It impressively navigates, accesses occlusion sites, and engages with effectiveness to aspirate clots, rapidly restoring blood flow in the patient’s brain—potentially a life-saving intervention.”
CereGlide 71 will be included in the next phase of the CERENOVUS EXCELLENT registry, a real-world registry focused on study of stroke-inducing blood clot removal via mechanical thrombectomy.
“CEREGLIDE 71 Intermediate Catheter is specifically designed to glide through challenging anatomical conditions,” said Mark Dickinson, Worldwide President, CERENOVUS. “Developed through robust research and clinical insights, our team designed a catheter for physicians that addresses unmet clinical needs by providing reliable trackability, durable delivery and the versatility for both direct aspiration and stent-retriever use—even in the most challenging anatomical conditions.”