Sam Brusco, Associate Editor02.05.24
Vektor Medical, a pioneer in non-invasive, artificial intelligence (AI)-based arrythmia analysis technology, has gained $16 million Series A investment. The funding round was led by Solas BioVentures and TVM Capital Life Science.
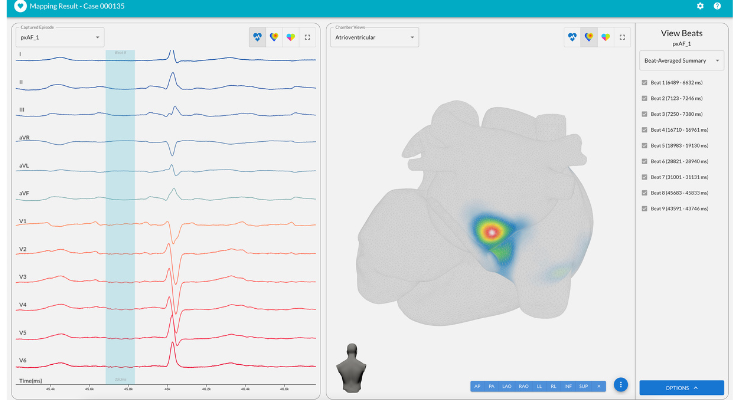
The company said the proceeds will support commercialization of vMap, an AI-based arrhythmia analysis tool that it hopes to transform how electrophysiologists (EPs) approach ablation procedures. Vektor said it will increase clinical support and drive additional clinical studies to bolster its expansion.
vMap is the only U.S. Food and Drug Administration (FDA)-cleared, non-invasive, AI-based solution to localize and analyze arrhythmia with a 12-lead ECG, the company said. vMap can help physicians better understand sources of arrhythmia and unlock actionable insights.
“The medical community is being confronted with the necessity for change. With an increasing atrial fibrillation (AF) patient population, outcomes must be improved while also enhancing procedure efficiency,” said Vektor CEO Rob Krummen. “vMap presents a solution to this challenge. Leading electrophysiologists (EPs) around the U.S. are utilizing vMap to streamline ablation procedures and improve patient outcomes. With the investment from Solas and TVM, we can expand access to this groundbreaking technology.”
In January 2024, the American Medical Association (AMA) CPT Editorial Panel approved a new Category III CPT code for the use of vMap.
“Vektor is addressing a massive problem in the cardiac arrhythmia space, giving millions of patients a chance to lead healthy lives free from this debilitating disease once again,” said Dr. David Adair, Managing Director of Solas BioVentures. “We are thrilled to partner with the team and support Vektor’s unwavering commitment to challenging the status quo across the entire cardiac arrhythmia pathway, making it easier and faster for physicians and health systems to access this breakthrough technology.”
The company said the proceeds will support commercialization of vMap, an AI-based arrhythmia analysis tool that it hopes to transform how electrophysiologists (EPs) approach ablation procedures. Vektor said it will increase clinical support and drive additional clinical studies to bolster its expansion.
vMap is the only U.S. Food and Drug Administration (FDA)-cleared, non-invasive, AI-based solution to localize and analyze arrhythmia with a 12-lead ECG, the company said. vMap can help physicians better understand sources of arrhythmia and unlock actionable insights.
“The medical community is being confronted with the necessity for change. With an increasing atrial fibrillation (AF) patient population, outcomes must be improved while also enhancing procedure efficiency,” said Vektor CEO Rob Krummen. “vMap presents a solution to this challenge. Leading electrophysiologists (EPs) around the U.S. are utilizing vMap to streamline ablation procedures and improve patient outcomes. With the investment from Solas and TVM, we can expand access to this groundbreaking technology.”
In January 2024, the American Medical Association (AMA) CPT Editorial Panel approved a new Category III CPT code for the use of vMap.
“Vektor is addressing a massive problem in the cardiac arrhythmia space, giving millions of patients a chance to lead healthy lives free from this debilitating disease once again,” said Dr. David Adair, Managing Director of Solas BioVentures. “We are thrilled to partner with the team and support Vektor’s unwavering commitment to challenging the status quo across the entire cardiac arrhythmia pathway, making it easier and faster for physicians and health systems to access this breakthrough technology.”