Michael Barbella, Managing Editor02.02.24
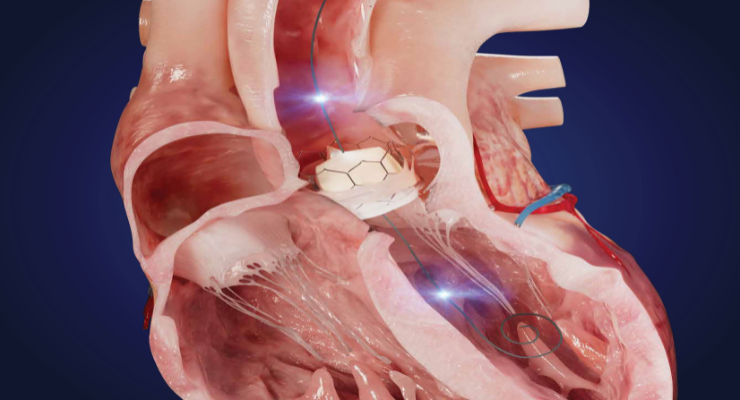
Xenter Inc. has unveiled its dual sensor investigational guidewire for transcatheter aortic valve replacement (TAVR) procedures. The product aims to collect an extensive range of crucial real-time data points to serve as an aortic regurgitation measurement (xAR) and clinical decision support tool, thus significantly enhancing para valvular leak (PVL) prediction during TAVR.
The guidewire’s functionality is designed to allow for comprehensive data collection and analysis and facilitates data necessary for artificial intelligence (AI) decision-making tools. The goal is to enable better-informed clinical decisions and improve patient outcomes. The guidewire is designed to operate seamlessly within a proprietary wireless ecosystem in the cardiac catheterization lab, making real-time data and analysis more accessible.
Xenter Chairman/CEO Rich Linder founded the company to help transform the collection of physical intelligence (PI) data into actionable insights, “We built Xenter to enable artificial intelligence tools to be utilized with ease and accuracy using new smart/wireless medical devices that transmit real-time PI data. Xenter is pioneering 'smart' medical devices and a wireless ecosystem that eliminate the capital equipment and mobile carts that clutter hospitals throughout the world,” he said. “Xenter is launching the XMD cloud that interfaces with hospital EHRs and enables AI-driven clinical decision support tools derived from real-time PI data.”
“An excellent example of real-time PI data collection is the TAVR SmartWire”, stated William A. Gray, M.D., System Chief of Cardiovascular Medicine at Mainline Healthcare in Philadelphia. “TAVR SmartWire will enable the real-time measurement of aortic and left ventricular pressure information in a single workhorse device that can be used to develop a measurement of aortic regurgitation (AR). We plan to clinically evaluate TAVR SmartWire and provide physicians with a real-time AR measurement called xAR.”
“From the beginning, we saw the need to innovate and engineer new solutions in cardiovascular medicine that can transform how information is collected during interventional procedures, including TAVR. I’m looking forward to seeing these solutions make their way into clinical research and to demonstrate the clinical utility of xAR in transcatheter aortic valve replacement,” said Martin B. Leon, M.D., Chairman Emeritus of the Cardiovascular Research Foundation in New York City, professor of Medicine at Columbia University/New York Presbyterian Hospital, and global study chair for TAVR SmartWire.
Xenter Inc. is a privately held company headquartered in Salt Lake City that is developing new wireless medical device technologies for providing real-time physical intelligence data.
The guidewire’s functionality is designed to allow for comprehensive data collection and analysis and facilitates data necessary for artificial intelligence (AI) decision-making tools. The goal is to enable better-informed clinical decisions and improve patient outcomes. The guidewire is designed to operate seamlessly within a proprietary wireless ecosystem in the cardiac catheterization lab, making real-time data and analysis more accessible.
Xenter Chairman/CEO Rich Linder founded the company to help transform the collection of physical intelligence (PI) data into actionable insights, “We built Xenter to enable artificial intelligence tools to be utilized with ease and accuracy using new smart/wireless medical devices that transmit real-time PI data. Xenter is pioneering 'smart' medical devices and a wireless ecosystem that eliminate the capital equipment and mobile carts that clutter hospitals throughout the world,” he said. “Xenter is launching the XMD cloud that interfaces with hospital EHRs and enables AI-driven clinical decision support tools derived from real-time PI data.”
“An excellent example of real-time PI data collection is the TAVR SmartWire”, stated William A. Gray, M.D., System Chief of Cardiovascular Medicine at Mainline Healthcare in Philadelphia. “TAVR SmartWire will enable the real-time measurement of aortic and left ventricular pressure information in a single workhorse device that can be used to develop a measurement of aortic regurgitation (AR). We plan to clinically evaluate TAVR SmartWire and provide physicians with a real-time AR measurement called xAR.”
“From the beginning, we saw the need to innovate and engineer new solutions in cardiovascular medicine that can transform how information is collected during interventional procedures, including TAVR. I’m looking forward to seeing these solutions make their way into clinical research and to demonstrate the clinical utility of xAR in transcatheter aortic valve replacement,” said Martin B. Leon, M.D., Chairman Emeritus of the Cardiovascular Research Foundation in New York City, professor of Medicine at Columbia University/New York Presbyterian Hospital, and global study chair for TAVR SmartWire.
Xenter Inc. is a privately held company headquartered in Salt Lake City that is developing new wireless medical device technologies for providing real-time physical intelligence data.