Sam Brusco, Associate Editor01.31.24
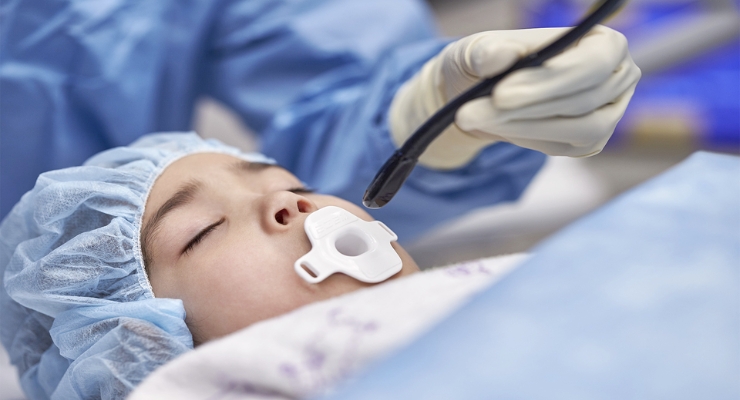
Philips has received U.S. Food and Drug Administration (FDA) 510(k) clearance for its latest TEE (transesophageal echocardiography) transducer, which was designed to serve more patients with improved overall comfort.
TEE provides detailed images of the heart and its internal structures. Philips said the quality of a 3D TEE image can help save lives in structural heart disease: It’s clarity and perspective exceed 2D alternatives. It’s also useful for minimally invasive heart surgeries like treating damaged heart valves and congenital heart defects.
Philips’ newly cleared X11-4t Mini 3D TEE transducer opens 3D TEE imaging to previously unaddressed patients, such as pediatric patients as small as five kilograms, adults at risk of complications, and complex cases in ICU patients where the transducer probe for 3D TEE was too large.

3D view of young pediatric patient heart view.
The transducer has a 35% smaller size and a pill-shaped form factor. The Mini Live 3D TEE solution uses the same hand control, procedure navigation techniques, and workflow as Philips’ EPIQ cardiac ultrasound systems. This requires minimal additional training on the new transducer for electrocardiographers, according to Philips.
The new TEE transducer is compatible with Philips’ EPIQ CVx and EchoNavigator image-guided therapy.
“As a pioneer and leading innovator in cardiac ultrasound, our 3D ultrasound technology plays a critical role in many cardiac procedures. But it was frustrating to know that there were still some patients who couldn’t benefit from this hugely beneficial approach to image the heart, and as a result, would often require a different, more invasive, treatment approach,” said David Handler, Philips’ VP and GM for Global Cardiology Ultrasound. “That’s why we’ve developed a new, even smaller mini 3D TEE transducer that can be used to help physicians serve a wider range of patients, from small children to fragile adults. With this innovation we can help reduce the need for general anesthesia and lower the risk of complications, meaning patients may recover faster from procedures and can be discharged sooner.”

3D view of adult left atrial appendage.
Philips said the X11-4t ultrasound transducer is scheduled for commercial availability in 2024 and is pending CE mark in Europe.
TEE provides detailed images of the heart and its internal structures. Philips said the quality of a 3D TEE image can help save lives in structural heart disease: It’s clarity and perspective exceed 2D alternatives. It’s also useful for minimally invasive heart surgeries like treating damaged heart valves and congenital heart defects.
Philips’ newly cleared X11-4t Mini 3D TEE transducer opens 3D TEE imaging to previously unaddressed patients, such as pediatric patients as small as five kilograms, adults at risk of complications, and complex cases in ICU patients where the transducer probe for 3D TEE was too large.

3D view of young pediatric patient heart view.
The transducer has a 35% smaller size and a pill-shaped form factor. The Mini Live 3D TEE solution uses the same hand control, procedure navigation techniques, and workflow as Philips’ EPIQ cardiac ultrasound systems. This requires minimal additional training on the new transducer for electrocardiographers, according to Philips.
The new TEE transducer is compatible with Philips’ EPIQ CVx and EchoNavigator image-guided therapy.
“As a pioneer and leading innovator in cardiac ultrasound, our 3D ultrasound technology plays a critical role in many cardiac procedures. But it was frustrating to know that there were still some patients who couldn’t benefit from this hugely beneficial approach to image the heart, and as a result, would often require a different, more invasive, treatment approach,” said David Handler, Philips’ VP and GM for Global Cardiology Ultrasound. “That’s why we’ve developed a new, even smaller mini 3D TEE transducer that can be used to help physicians serve a wider range of patients, from small children to fragile adults. With this innovation we can help reduce the need for general anesthesia and lower the risk of complications, meaning patients may recover faster from procedures and can be discharged sooner.”

3D view of adult left atrial appendage.
Philips said the X11-4t ultrasound transducer is scheduled for commercial availability in 2024 and is pending CE mark in Europe.