Sam Brusco, Associate Editor01.31.24
Boston Scientific has earned U.S. Food and Drug Administration (FDA) approval for its Farapulse pulsed field ablation (PFA) system.
The technology is indicated for isolation of pulmonary veins to treat drug-refractory, recurrent, symptomatic, paroxysmal atrial fibrillation (AFib). PFA is a new alternative to standard-of-case thermal ablation treatment, where a catheter is guided to the heart’s interior and generates extreme hot or cold to destroy targeted areas associated with arrhythmias.
Farapulse PFA instead uses tissue-selective, non-thermal electric fields to ablate heart tissue and avoid damage to surrounding anatomy. 12-month data from Boston Scientific’s pivotal ADVENT trial found the device matched the safety and efficacy of conventional thermal ablation, with shorter ablation times and a fast learning curve for physicians.
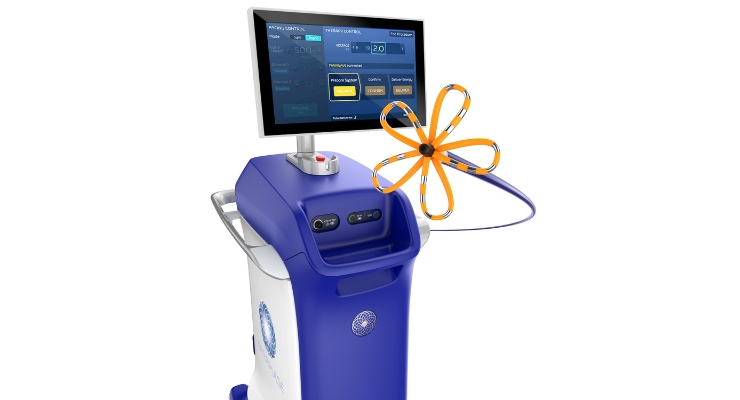
The system consists of the FaraWave ablation catheter, FaraStar ablation generator, and FaraDrive steerable sheath. These are complemented by the VersaCross Connect access solution for the steerable sheath to offer safe, efficient access to the left side of the heart during procedures. FaraWave can treat a range of pulmonary vein anatomies using an over-the-wire catheter with variable basket and flower shapes, according to Boston Scientific.
"The approval of the FARAPULSE PFA System marks an important milestone for the millions of people living with paroxysmal AF and is an incredible opportunity to bring the first PFA system designed and built solely for this type of ablation therapy to physicians in the U.S.," said Nick Spadea-Anello, president, Electrophysiology, Boston Scientific. "A high bar has been set by performance of the system in clinical and commercial settings—where more than 40,000 patients have been treated to date—and we look forward to continuing to lead the way with this differentiated technology in the growing PFA space."
Boston Scientific finished enrollment in the first phase of the ADVANTAGE AF clinical trial in Q3 2023, which is studying the system to treat patients with drug-refractory, symptomatic, persistent AFib, and began enrollment in a second phase of the study to evaluate safety and effectiveness of adjunctive use of the FaraPoint PFA Catheter for cavotricuspid isthmus (CTI) ablations, a procedure used to treat atrial flutter.
The company also recently commenced the AVANT GUARD clinical trial to evaluate the safety and efficacy of the system as a first-line treatment for persistent AF compared to anti-arrhythmic drug therapy.
The technology is indicated for isolation of pulmonary veins to treat drug-refractory, recurrent, symptomatic, paroxysmal atrial fibrillation (AFib). PFA is a new alternative to standard-of-case thermal ablation treatment, where a catheter is guided to the heart’s interior and generates extreme hot or cold to destroy targeted areas associated with arrhythmias.
Farapulse PFA instead uses tissue-selective, non-thermal electric fields to ablate heart tissue and avoid damage to surrounding anatomy. 12-month data from Boston Scientific’s pivotal ADVENT trial found the device matched the safety and efficacy of conventional thermal ablation, with shorter ablation times and a fast learning curve for physicians.
The system consists of the FaraWave ablation catheter, FaraStar ablation generator, and FaraDrive steerable sheath. These are complemented by the VersaCross Connect access solution for the steerable sheath to offer safe, efficient access to the left side of the heart during procedures. FaraWave can treat a range of pulmonary vein anatomies using an over-the-wire catheter with variable basket and flower shapes, according to Boston Scientific.
"The approval of the FARAPULSE PFA System marks an important milestone for the millions of people living with paroxysmal AF and is an incredible opportunity to bring the first PFA system designed and built solely for this type of ablation therapy to physicians in the U.S.," said Nick Spadea-Anello, president, Electrophysiology, Boston Scientific. "A high bar has been set by performance of the system in clinical and commercial settings—where more than 40,000 patients have been treated to date—and we look forward to continuing to lead the way with this differentiated technology in the growing PFA space."
Boston Scientific finished enrollment in the first phase of the ADVANTAGE AF clinical trial in Q3 2023, which is studying the system to treat patients with drug-refractory, symptomatic, persistent AFib, and began enrollment in a second phase of the study to evaluate safety and effectiveness of adjunctive use of the FaraPoint PFA Catheter for cavotricuspid isthmus (CTI) ablations, a procedure used to treat atrial flutter.
The company also recently commenced the AVANT GUARD clinical trial to evaluate the safety and efficacy of the system as a first-line treatment for persistent AF compared to anti-arrhythmic drug therapy.
More Activity in Pulsed Field Ablation
- Medtronic’s PulseSelect PFA was FDA approved to treat both paroxysmal and persistent AFib in December 2023.
- Abbott completed the first global procedures with its Volt PFA system earlier this month.