Sam Brusco, Associate Editor01.30.24
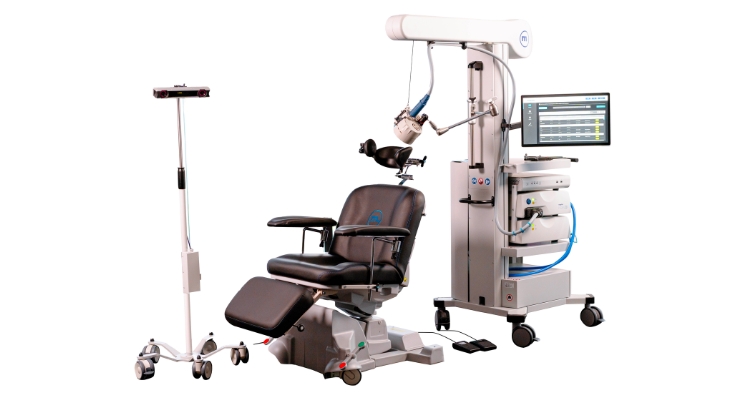
Magstim gained U.S. Food and Drug Administration (FDA) clearance for its Horizon 3.0 with StimGuide Pro. The transcranial magnetic stimulation (TMS) technology treats those diagnosed with depressive or obsessive-compulsive conditions.
TMS is non-invasive brain stimulation that leverages an electromagnetic coil to stimulate nerve cells in the brain region involved in mood control and depression.
Horizon 3.0 with StimGuide Pro builds on Magstim’s H 3.0 platform, adding advanced camera technology and software for precise treatment targeting. Patient treatments and technology are centralized on one screen to reduce complexity so healthcare practitioners can focus on care, according to the company.
StimGuide Pro advanced navigation was designed to improve coil positioning. The new camera and navigation tools, said Magstim, ensure delivery of magnetic pulses to targeted brain regions. Magstim Connect also lets multiple compatible systems be managed from a central system.
“With just one screen, this system is simple to use and navigate,” said Anthony Baldwin Polizzi, Senior TMS Technician, My Psychiatrist, Florida. “The new Bluetooth anatomical pointer makes it easy to map and treat patients. I like it a lot.”
Magstim built the first TMS system over 30 years ago. The company said it’s been used in over 16,000 neuroscientific and clinical research studies.
“We are passionate about helping patients worldwide to improve their mental health,” said Ronnie Stolec-Campo, CEO, Magstim. “We worked with leading psychiatrists, clinicians, and researchers to develop this system, enabling advanced treatments and simplified practice workflows. TMS is life-saving technology that provides non-pharmacological, non-invasive treatments.”
In March 2023, Magstim earned FDA clearance for Horizon 3.0 to treat adults diagnosed with obsessive compulsive disorder.
TMS is non-invasive brain stimulation that leverages an electromagnetic coil to stimulate nerve cells in the brain region involved in mood control and depression.
Horizon 3.0 with StimGuide Pro builds on Magstim’s H 3.0 platform, adding advanced camera technology and software for precise treatment targeting. Patient treatments and technology are centralized on one screen to reduce complexity so healthcare practitioners can focus on care, according to the company.
StimGuide Pro advanced navigation was designed to improve coil positioning. The new camera and navigation tools, said Magstim, ensure delivery of magnetic pulses to targeted brain regions. Magstim Connect also lets multiple compatible systems be managed from a central system.
“With just one screen, this system is simple to use and navigate,” said Anthony Baldwin Polizzi, Senior TMS Technician, My Psychiatrist, Florida. “The new Bluetooth anatomical pointer makes it easy to map and treat patients. I like it a lot.”
Magstim built the first TMS system over 30 years ago. The company said it’s been used in over 16,000 neuroscientific and clinical research studies.
“We are passionate about helping patients worldwide to improve their mental health,” said Ronnie Stolec-Campo, CEO, Magstim. “We worked with leading psychiatrists, clinicians, and researchers to develop this system, enabling advanced treatments and simplified practice workflows. TMS is life-saving technology that provides non-pharmacological, non-invasive treatments.”
In March 2023, Magstim earned FDA clearance for Horizon 3.0 to treat adults diagnosed with obsessive compulsive disorder.