Sam Brusco, Associate Editor01.15.24
Last month, ResMed issued a safety notice concerning its AirFit and AirTouch CPAP masks about potential magnetic interference when the masks’ magnets are near some implants and devices in the body.
Now, the U.S. Food and Drug Administration (FDA) has identified this safety issue as a Class I recall.
The AirFit and AirTouch masks are non-continuous ventilatory devices, intended to be used by patients weighing over 66 pounds who have been prescribed non-invasive positive airway pressure (PAP) therapy such as CPAP or bi-level therapy. The masks are meant for reuse by one person at home or by multiple people in hospitals.
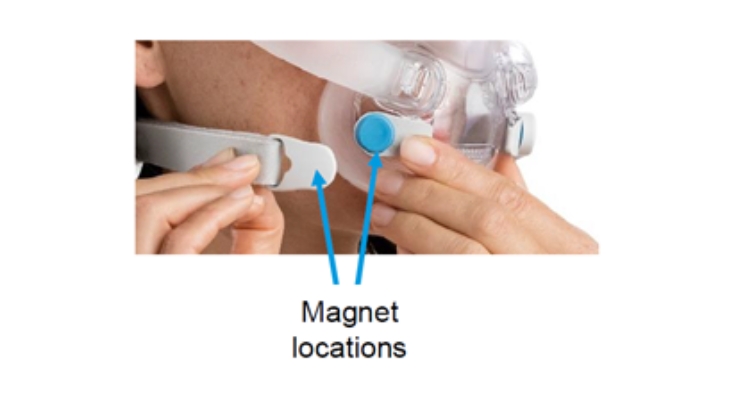
The devices have magnets on the lower headgear straps and frame connections of CPAP masks. These magnets are there to make wearing the mask more comfortable.
Under certain circumstances when the magnet is less than two inches from certain implants and devices, interference can occur and disrupt their function or position. The existing label advises keeping magnets two inches away from affected devices but it doesn’t list the all specific ones that could be affected, the FDA said.
ResMed is recalling all CPAP masks with magnets to update the labels, as well as add more warnings and information to guide safe usage when using masks with magnets. So far, ResMed has reported six injuries due to the magnetic interference issue, with no reports of death.
The company’s Important Medical Device Advisory urged customers to keep the magnets six inches away from implants or devices that could be affected by magnetic interference, and reach out to their doctor or device manufacturer about potential negative effects of magnetic fields on their device.
Contraindicated devices at risk of magnetic interference include:
Now, the U.S. Food and Drug Administration (FDA) has identified this safety issue as a Class I recall.
The AirFit and AirTouch masks are non-continuous ventilatory devices, intended to be used by patients weighing over 66 pounds who have been prescribed non-invasive positive airway pressure (PAP) therapy such as CPAP or bi-level therapy. The masks are meant for reuse by one person at home or by multiple people in hospitals.
The devices have magnets on the lower headgear straps and frame connections of CPAP masks. These magnets are there to make wearing the mask more comfortable.
Under certain circumstances when the magnet is less than two inches from certain implants and devices, interference can occur and disrupt their function or position. The existing label advises keeping magnets two inches away from affected devices but it doesn’t list the all specific ones that could be affected, the FDA said.
ResMed is recalling all CPAP masks with magnets to update the labels, as well as add more warnings and information to guide safe usage when using masks with magnets. So far, ResMed has reported six injuries due to the magnetic interference issue, with no reports of death.
The company’s Important Medical Device Advisory urged customers to keep the magnets six inches away from implants or devices that could be affected by magnetic interference, and reach out to their doctor or device manufacturer about potential negative effects of magnetic fields on their device.
Contraindicated devices at risk of magnetic interference include:
- Active medical implants (pacemakers, ICDs, neurostimulators, CSF shunts, insulin/infusion pumps)
- Metallic implants/objects with ferromagnetic material (aneurysm clips/flow disruption devices, embolic coils, stents, valves, electrodes, implants to restore hearing or balance with implanted magnets, ocular implants, metallic splinters in the eye)