Michael Barbella, Managing Editor01.12.24
The American Medical Association (AMA) CPT Editorial Panel has approved a new Category III CPT code for the use of Vektor Medical's vMap’s noninvasive arrhythmia analysis.
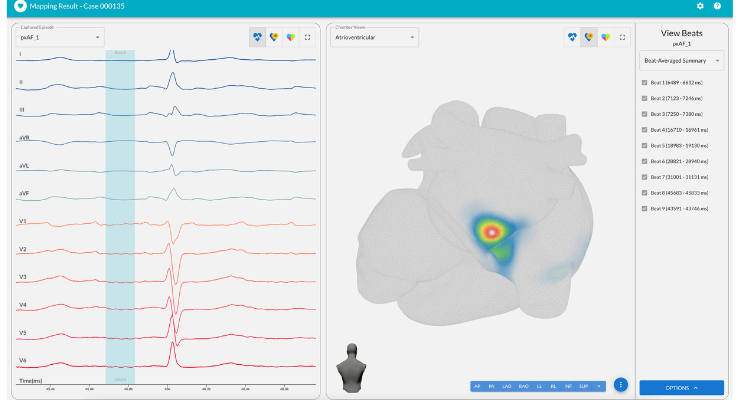
vMap is a U.S. Food and Drug Administration (FDA)-cleared, artificial intelligence (AI)-based arrhythmia analysis system that has been shown to significantly improve cardiac ablation outcomes, enhance procedural efficiency, and optimize workflows. This solution, utilizing an AI-derived arrhythmia analysis, delivers valuable insights to help in treatment decision-making, optimize patient triage, and streamline the planning and execution of catheter ablation procedures.
"We are delighted to have the CPT code for vMap approved. This is a significant and pivotal milestone for the company, and we are deeply honored to receive the AMA's confirmation that vMap provides unique value deserving of appropriate recognition,” Vektor Medical CEO Rob Krummen stated. “This code will further enable the positive impact vMap is able to have on patient outcomes and procedural efficiencies for the millions of people suffering from arrhythmias. This achievement not only signifies a major step forward for Vektor Medical, but also underscores the importance and recognition of innovative technologies to enable healthcare transformation.”
The AMA CPT Editorial Panel has published the code describing vMap’s noninvasive arrhythmia analysis derived from quantitative computational cardiac arrhythmia simulations ahead of its expected July 1 implementation.
Latham & Watkins LLP provided reimbursement counsel to Vektor Medical throughout the CPT process.
Arrhythmias are a mounting concern in the United States and globally. Although cardiac ablation stands as the most effective treatment for cardiac arrhythmias, its utilization remains starkly low. vMap has been shown in clinical studies to reduce risk, cost, and procedure time; to improve success rates of ablation therapy; and to broaden the scope of patients benefiting from ablation. vMap is intended to pave the way for a more effective, accessible, and patient-centric approach to tackling this urgent global health issue.
vMap is the only FDA-cleared, noninvasive, AI-based solution for mapping arrhythmias, utilizing just a 12-lead ECG. vMap leverages AI-based algorithms to locate arrhythmia sources for focal and fibrillation-type arrhythmias, including atrial fibrillation. By unlocking actionable insights contained within a standard 12-lead ECG, vMap empowers physicians to rapidly and accurately locate arrhythmia source locations. Supported by clinical evidence, vMap not only elevates cardiac ablation outcomes but also reduces procedure time, optimizing the entire arrhythmia care pathway.
Headquartered in San Diego, Vektor Medical aims to revolutionize arrhythmia care for millions affected by atrial fibrillation and other arrhythmias. Through its AI-based, noninvasive arrhythmia analysis technology (vMap), the company aspires to improve cardiac ablation outcomes, optimize workflows, and increase procedural efficiency.
vMap is a U.S. Food and Drug Administration (FDA)-cleared, artificial intelligence (AI)-based arrhythmia analysis system that has been shown to significantly improve cardiac ablation outcomes, enhance procedural efficiency, and optimize workflows. This solution, utilizing an AI-derived arrhythmia analysis, delivers valuable insights to help in treatment decision-making, optimize patient triage, and streamline the planning and execution of catheter ablation procedures.
"We are delighted to have the CPT code for vMap approved. This is a significant and pivotal milestone for the company, and we are deeply honored to receive the AMA's confirmation that vMap provides unique value deserving of appropriate recognition,” Vektor Medical CEO Rob Krummen stated. “This code will further enable the positive impact vMap is able to have on patient outcomes and procedural efficiencies for the millions of people suffering from arrhythmias. This achievement not only signifies a major step forward for Vektor Medical, but also underscores the importance and recognition of innovative technologies to enable healthcare transformation.”
The AMA CPT Editorial Panel has published the code describing vMap’s noninvasive arrhythmia analysis derived from quantitative computational cardiac arrhythmia simulations ahead of its expected July 1 implementation.
Latham & Watkins LLP provided reimbursement counsel to Vektor Medical throughout the CPT process.
Arrhythmias are a mounting concern in the United States and globally. Although cardiac ablation stands as the most effective treatment for cardiac arrhythmias, its utilization remains starkly low. vMap has been shown in clinical studies to reduce risk, cost, and procedure time; to improve success rates of ablation therapy; and to broaden the scope of patients benefiting from ablation. vMap is intended to pave the way for a more effective, accessible, and patient-centric approach to tackling this urgent global health issue.
vMap is the only FDA-cleared, noninvasive, AI-based solution for mapping arrhythmias, utilizing just a 12-lead ECG. vMap leverages AI-based algorithms to locate arrhythmia sources for focal and fibrillation-type arrhythmias, including atrial fibrillation. By unlocking actionable insights contained within a standard 12-lead ECG, vMap empowers physicians to rapidly and accurately locate arrhythmia source locations. Supported by clinical evidence, vMap not only elevates cardiac ablation outcomes but also reduces procedure time, optimizing the entire arrhythmia care pathway.
Headquartered in San Diego, Vektor Medical aims to revolutionize arrhythmia care for millions affected by atrial fibrillation and other arrhythmias. Through its AI-based, noninvasive arrhythmia analysis technology (vMap), the company aspires to improve cardiac ablation outcomes, optimize workflows, and increase procedural efficiency.