Sam Brusco, Associate Editor11.16.23
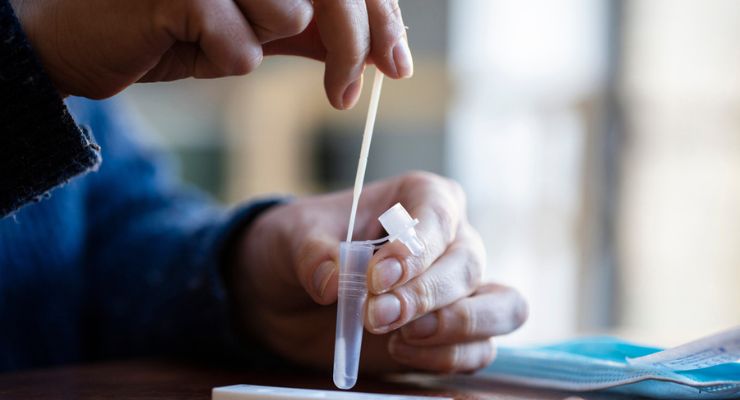
LetsGetChecked has earned De Novo authorization from the U.S. Food and Drug Administration for Simple 2, the company’s at-home collection system to test for chlamydia (CT) and gonorrhea (NG). The direct-to-consumer test can be purchased without a prescription.
This de novo marks the first FDA-authorized test with home sample collection for a sexually transmitted disease other than HIV and first home sample collection system with retail and online access to the kit. The de novo has created a new regulatory classification as a result. The authorization of LetsGetChecked’s end-to-end testing system includes manufacturing, logistics, lab analysis, and healthcare support.
The FDA evaluated data from LetsGetChecked demonstrating lay users can safely use the kit, have general understanding of the results and any needed follow up actions, and validate their home collection kits with the intended test. The collection kit is validated online and the user fills out a health questionnaire for a healthcare provider to look over.
“This authorization marks an important public health milestone, giving patients more information about their health from the privacy of their own home,” said Jeff Shuren, M.D., J.D., director of the FDA’s Center for Devices and Radiological Health, said in an FDA statement. “We are eager to continue supporting greater consumer access to diagnostic tests, which helps further our goal of bringing more health care into the home.”
The Simple 2 test is performed through vaginal swab or urine sample for people 18 and over, using Hologic’s Aptima collection devices. The samples are then analyzed in LetsGetChecked’s lab, which is CLIA- and CAP-certified in the U.S. to test using the Hologic Aptima Combo 2 assay (for CT/NG).
“We are honored to receive this landmark De Novo classification and authorization from FDA for our chlamydia and gonorrhea testing system, which demonstrates the value of our robust end-to-end at-home care system,” LetsGetChecked’s founder and CEO Peter Foley told the press. “Our vertically integrated platform, coupled with the authorization of our testing solution will empower individuals to proactively manage their health from home. We greatly appreciate FDA’s collaboration throughout this process.”
“We are proud to support LetsGetChecked on this first-ever FDA-authorized at-home collection kit for chlamydia and gonorrhea, which uses our best-in-class technology,” added Hologic Diagnostics Solutions’ president Jennifer Schneiders, Ph.D. “Our priority as an organization is to ensure people have access to the highest standards of care to safeguard their health. As STIs continue to rise at alarming rates, we are committed to innovative testing strategies that bring additional testing options to people everywhere.”
This de novo marks the first FDA-authorized test with home sample collection for a sexually transmitted disease other than HIV and first home sample collection system with retail and online access to the kit. The de novo has created a new regulatory classification as a result. The authorization of LetsGetChecked’s end-to-end testing system includes manufacturing, logistics, lab analysis, and healthcare support.
The FDA evaluated data from LetsGetChecked demonstrating lay users can safely use the kit, have general understanding of the results and any needed follow up actions, and validate their home collection kits with the intended test. The collection kit is validated online and the user fills out a health questionnaire for a healthcare provider to look over.
“This authorization marks an important public health milestone, giving patients more information about their health from the privacy of their own home,” said Jeff Shuren, M.D., J.D., director of the FDA’s Center for Devices and Radiological Health, said in an FDA statement. “We are eager to continue supporting greater consumer access to diagnostic tests, which helps further our goal of bringing more health care into the home.”
The Simple 2 test is performed through vaginal swab or urine sample for people 18 and over, using Hologic’s Aptima collection devices. The samples are then analyzed in LetsGetChecked’s lab, which is CLIA- and CAP-certified in the U.S. to test using the Hologic Aptima Combo 2 assay (for CT/NG).
“We are honored to receive this landmark De Novo classification and authorization from FDA for our chlamydia and gonorrhea testing system, which demonstrates the value of our robust end-to-end at-home care system,” LetsGetChecked’s founder and CEO Peter Foley told the press. “Our vertically integrated platform, coupled with the authorization of our testing solution will empower individuals to proactively manage their health from home. We greatly appreciate FDA’s collaboration throughout this process.”
“We are proud to support LetsGetChecked on this first-ever FDA-authorized at-home collection kit for chlamydia and gonorrhea, which uses our best-in-class technology,” added Hologic Diagnostics Solutions’ president Jennifer Schneiders, Ph.D. “Our priority as an organization is to ensure people have access to the highest standards of care to safeguard their health. As STIs continue to rise at alarming rates, we are committed to innovative testing strategies that bring additional testing options to people everywhere.”