Sam Brusco, Associate Editor10.23.23
Medtronic has earned U.S. Food and Drug Administration (FDA) approval for its Aurora EV-ICD MRI SureScan (extravascular implantable cardioverter-defibrillator) and Epsila EV MRI SureScan defibrillation lead to treat dangerously fast heart rhythms that can cause sudden cardiac arrest (SCA).
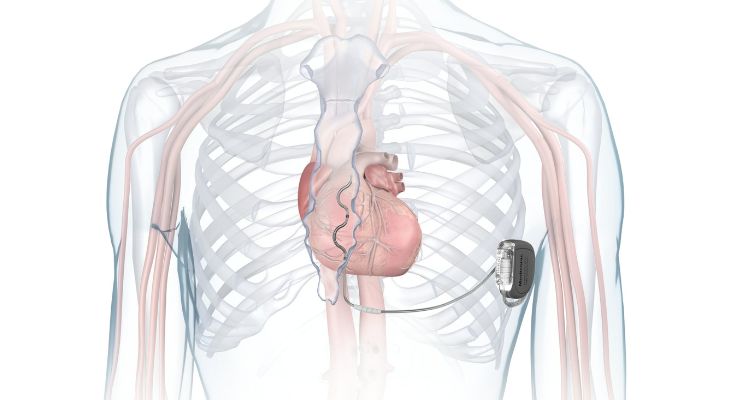
According to Medtronic, Aurora is the first of its kind to have the same benefits as traditional, transvenous ICDs with a lead placed under the breastbone, outside of the heart and veins. It provides defibrillation, anti-tachycardia pacing (ATP), and back-up (pause-prevention) pacing therapies via a device similar in size, shape, and longevity to transvenous ICDs. It earned CE mark approval in February of this year.
FDA approval also covers Aurora’s procedure implant tools. The system will become available on a limited basis in the U.S. in the coming weeks, Medtronic said.
 "This FDA approval paves the way for patients to have a better overall experience with ICD therapy," Alan Cheng, MD, chief medical officer of Medtronic’s Cardiac Rhythm Management business told the press. "ICDs remain the gold standard for prevention of sudden cardiac death, and while the subcutaneous ICD avoids certain complications associated with transvenous defibrillators, it has limitations that may affect a patient's comfort and quality-of-life. With the Aurora EV-ICD system, patients can benefit from the only ICD placed outside the vascular space that provides ATP and back-up pacing, in a device that is nearly half the size and with 60% greater projected battery longevity compared to the competitor's subcutaneous ICD."
"This FDA approval paves the way for patients to have a better overall experience with ICD therapy," Alan Cheng, MD, chief medical officer of Medtronic’s Cardiac Rhythm Management business told the press. "ICDs remain the gold standard for prevention of sudden cardiac death, and while the subcutaneous ICD avoids certain complications associated with transvenous defibrillators, it has limitations that may affect a patient's comfort and quality-of-life. With the Aurora EV-ICD system, patients can benefit from the only ICD placed outside the vascular space that provides ATP and back-up pacing, in a device that is nearly half the size and with 60% greater projected battery longevity compared to the competitor's subcutaneous ICD."
Aurora is implanted in the left mid-axillary region, below the left armpit. The Epsila EV defibrillation lead is placed under the sternum outside the heart and veins to avoid the vascular injuries and vessel occlusions related to transvenous leads.
In the pivotal study, the device's effectiveness in delivering defibrillation therapy at implant was reportedly 98.7%, and there we no major intraprocedural complications, nor any unique complications observed related to the EV ICD procedure or system compared to transvenous and subcutaneous ICDs.
Medtronic said it will gather real-world performance and safety data on Aurora in the Enlighten global post-approval registry, a prospective, non-randomized, observational, multicenter study, expected to last five years and enroll about 1,000 patients.
According to Medtronic, Aurora is the first of its kind to have the same benefits as traditional, transvenous ICDs with a lead placed under the breastbone, outside of the heart and veins. It provides defibrillation, anti-tachycardia pacing (ATP), and back-up (pause-prevention) pacing therapies via a device similar in size, shape, and longevity to transvenous ICDs. It earned CE mark approval in February of this year.
FDA approval also covers Aurora’s procedure implant tools. The system will become available on a limited basis in the U.S. in the coming weeks, Medtronic said.

Aurora is implanted in the left mid-axillary region, below the left armpit. The Epsila EV defibrillation lead is placed under the sternum outside the heart and veins to avoid the vascular injuries and vessel occlusions related to transvenous leads.
In the pivotal study, the device's effectiveness in delivering defibrillation therapy at implant was reportedly 98.7%, and there we no major intraprocedural complications, nor any unique complications observed related to the EV ICD procedure or system compared to transvenous and subcutaneous ICDs.
Medtronic said it will gather real-world performance and safety data on Aurora in the Enlighten global post-approval registry, a prospective, non-randomized, observational, multicenter study, expected to last five years and enroll about 1,000 patients.