Sam Brusco, Associate Editor10.12.23
Medtronic has gained CE mark approval for its newest-generation Evolut FX transcatheter aortic valve implantation (TAVI) system.
Evolut FX is the latest of the company’s CoreValve/Evolut TAVI platform, and according to Medtronic is the only platform to show durability advantage over surgical aortic valve replacement (SAVR) at five and 10 years in a randomized trial.
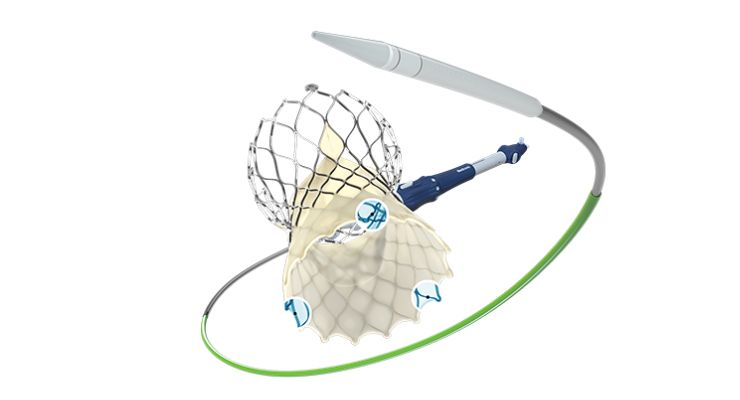
The company’s TAVI system features a supra-annular, self-expanding valve design. The now fourth-generation Evolut has gold markers built in the frame to help surgeons directly visualize depth of valve leaflet orientation during procedures. A redesigned delivery catheter, Medtronic says, creates a smoother insertion profile and more stable system.
Evolut FX is now indicated for severe aortic stenosis in adults across all risk categories in the EU. In the U.S., it’s indicated for symptomatic, severe aortic stenosis in adults across all risk categories.
“This exciting milestone helps us continually enhance a trusted platform to better respond to clinicians' needs making TAVI procedures easier to visualize and more predictable for heart teams,” VP and chief medical officer of Medtronic’s Coronary & Renal Denervation business Jeffrey Popma, MD told the press. “Receiving CE mark for the Evolut FX system highlights our commitment to providing minimally invasive treatment options globally for patients experiencing severe aortic stenosis.”
Evolut FX is the latest of the company’s CoreValve/Evolut TAVI platform, and according to Medtronic is the only platform to show durability advantage over surgical aortic valve replacement (SAVR) at five and 10 years in a randomized trial.
The company’s TAVI system features a supra-annular, self-expanding valve design. The now fourth-generation Evolut has gold markers built in the frame to help surgeons directly visualize depth of valve leaflet orientation during procedures. A redesigned delivery catheter, Medtronic says, creates a smoother insertion profile and more stable system.
Evolut FX is now indicated for severe aortic stenosis in adults across all risk categories in the EU. In the U.S., it’s indicated for symptomatic, severe aortic stenosis in adults across all risk categories.
“This exciting milestone helps us continually enhance a trusted platform to better respond to clinicians' needs making TAVI procedures easier to visualize and more predictable for heart teams,” VP and chief medical officer of Medtronic’s Coronary & Renal Denervation business Jeffrey Popma, MD told the press. “Receiving CE mark for the Evolut FX system highlights our commitment to providing minimally invasive treatment options globally for patients experiencing severe aortic stenosis.”