Sam Brusco, Associate Editor10.09.23
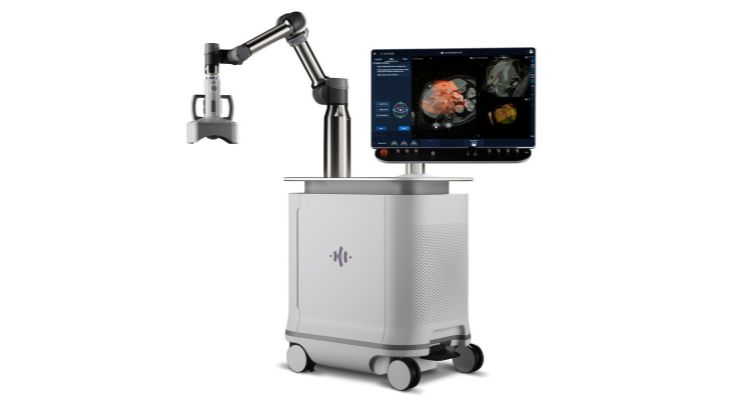
HistoSonics has gained U.S. Food and Drug Administration (FDA) De Novo clearance for its Edison histotripsy system, making it the first of its type available in the U.S. The science of histotripsy uses focused sound energy to produce controlled acoustic cavitation that mechanically destroys and liquifies targeted liver tissue, including tumors, at sub-cellular levels.
The system is indicated for non-invasive destruction of liver tumors—including unresectable tumors—with a non-thermal, mechanical focused ultrasound process. The company believes using Edison can help the treatment site recover and absorb quickly. The platform also allows monitoring of destruction of tissue under continuous, real-time visualization and control.
Edison gained FDA breakthrough device designation in October 2021.
The De Novo authorization is based (in part) on #HOPE4LIVER trials data in the U.S. and Europe. The trial data found histotripsy’s primary safety and efficacy to treat secondary liver tumors. As recently presented at the CIRSE Congress in Copenhagen, a technical success rate of 95.5% was achieved, showing physicians can precisely target and destroy liver tissue and unresectable liver tumors.
Further, only three procedure-related CTCAE Grade 3 or higher adverse events through 30 days post-histotripsy were seen across all 44 subjects treated, representing a complication rate of 6.8% with each event being common to focal liver therapies and not specific to histotripsy.
“This is HistoSonics’ most meaningful milestone to date and represents over two-decades of tireless efforts, from its inception at the University of Michigan in 2001, overcoming what was once thought to be impossible—integrating the many complexities of histotripsy into a completely non-invasive clinical platform,” HistoSonics’ president and CEO Mike Blue told the press. “We have been thoughtfully adding professionals with deep domain experience in operations, market development and education and are prepared to begin scheduling physician training immediately. This is a fantastic day for patients who will benefit from the novel advantages of histotripsy, and I commend the FDA for working so expeditiously with us throughout the review process.”
The company also achieve FDA approval for the sponsored #HOP4KIDNEY trial earlier this year.
The system is indicated for non-invasive destruction of liver tumors—including unresectable tumors—with a non-thermal, mechanical focused ultrasound process. The company believes using Edison can help the treatment site recover and absorb quickly. The platform also allows monitoring of destruction of tissue under continuous, real-time visualization and control.
Edison gained FDA breakthrough device designation in October 2021.
The De Novo authorization is based (in part) on #HOPE4LIVER trials data in the U.S. and Europe. The trial data found histotripsy’s primary safety and efficacy to treat secondary liver tumors. As recently presented at the CIRSE Congress in Copenhagen, a technical success rate of 95.5% was achieved, showing physicians can precisely target and destroy liver tissue and unresectable liver tumors.
Further, only three procedure-related CTCAE Grade 3 or higher adverse events through 30 days post-histotripsy were seen across all 44 subjects treated, representing a complication rate of 6.8% with each event being common to focal liver therapies and not specific to histotripsy.
“This is HistoSonics’ most meaningful milestone to date and represents over two-decades of tireless efforts, from its inception at the University of Michigan in 2001, overcoming what was once thought to be impossible—integrating the many complexities of histotripsy into a completely non-invasive clinical platform,” HistoSonics’ president and CEO Mike Blue told the press. “We have been thoughtfully adding professionals with deep domain experience in operations, market development and education and are prepared to begin scheduling physician training immediately. This is a fantastic day for patients who will benefit from the novel advantages of histotripsy, and I commend the FDA for working so expeditiously with us throughout the review process.”
The company also achieve FDA approval for the sponsored #HOP4KIDNEY trial earlier this year.