Sam Brusco, Associate Editor09.26.23
Senseonics, a company developing and manufacturing long-term, implantable continuous glucose monitoring (CGM) systems, completed its ENHANCE pivotal clinical study adult cohort after the recently completed 365-day visit for the final study patient.
“We are pleased that all patients have completed the testing for the adult arm of the ENHANCE clinical study. Demonstration of safe and accurate performance of Eversense for 365 days in this study would further validate its ability to facilitate more effective and longer-term management of diabetes. This represents a major milestone for Senseonics as we work towards offering the differentiated benefits of implantable CGM for one full year with just a single sensor,” Senseonics’ chief medical officer Francine Kaufman, MD told the press. “We appreciate the efforts of the participating investigators and patients and look forward to analyzing the data, preparing the submission and working with the FDA with the goal to secure approval for the planned 365-day Eversense system in the coming quarters.”
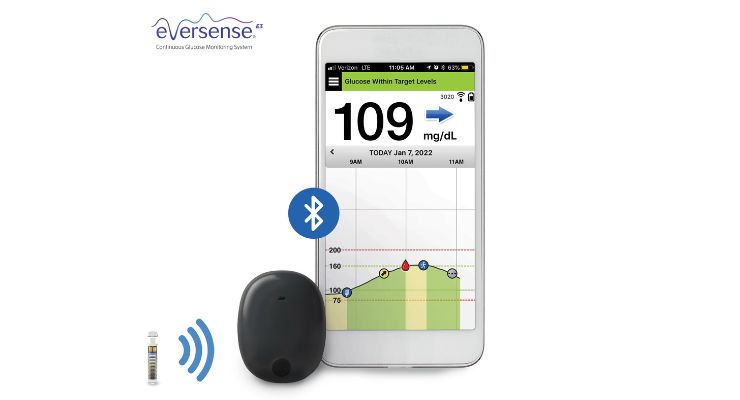
The ENHANCE study aims to evaluate Senseonics’ Eversense iCGM system for a year. More than 165 adults were implanted with Eversense systems in four U.S. centers.
Enrollment for the 365-day sensor was completed in September 2022. Data gathered from this study was also used to support U.S. Food and Drug Administration (FDA) submission for the integrated continuous glucose monitoring (iCGM) designation earlier this year.
An investigational device exemption (IDE) supplement was approved for the study’s expansion to allow for patients aged 14 to 18, and the first pediatric study participants were enrolled in Q2 2023.
“We are pleased that all patients have completed the testing for the adult arm of the ENHANCE clinical study. Demonstration of safe and accurate performance of Eversense for 365 days in this study would further validate its ability to facilitate more effective and longer-term management of diabetes. This represents a major milestone for Senseonics as we work towards offering the differentiated benefits of implantable CGM for one full year with just a single sensor,” Senseonics’ chief medical officer Francine Kaufman, MD told the press. “We appreciate the efforts of the participating investigators and patients and look forward to analyzing the data, preparing the submission and working with the FDA with the goal to secure approval for the planned 365-day Eversense system in the coming quarters.”
The ENHANCE study aims to evaluate Senseonics’ Eversense iCGM system for a year. More than 165 adults were implanted with Eversense systems in four U.S. centers.
Enrollment for the 365-day sensor was completed in September 2022. Data gathered from this study was also used to support U.S. Food and Drug Administration (FDA) submission for the integrated continuous glucose monitoring (iCGM) designation earlier this year.
An investigational device exemption (IDE) supplement was approved for the study’s expansion to allow for patients aged 14 to 18, and the first pediatric study participants were enrolled in Q2 2023.