Sam Brusco, Associate Editor09.21.23
Medtronic has gained CE mark approval for its new disposable Simplera continuous glucose monitor (CGM), which features a two-step insertion process.
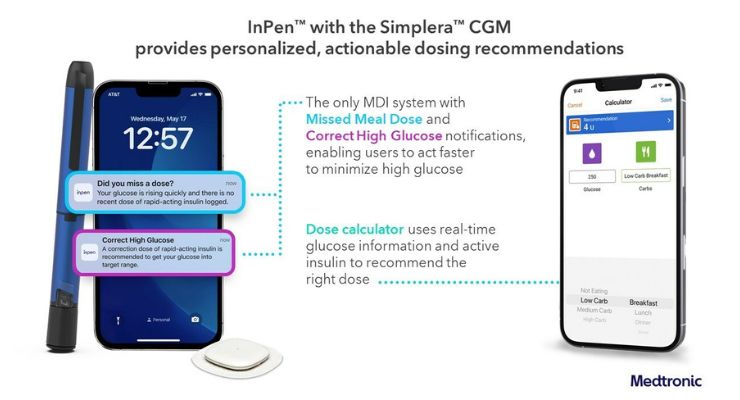
Simplera touts Medtronic’s newest, no-fingerstick sensor (which requires no over tape) and integrates with InPen—the company’s smart insulin pen that leverages an adjustable algorithm to add real-time, personalized dosing guidance for diabetes management.
"Patients with diabetes can get overwhelmed by the sheer number of decisions they need to make on a daily basis. As a physician, I appreciate the ability to introduce this solution by Medtronic as it provides real-time, personalized guidance to help patients stay in range. For instance, when it detects someone is consuming a meal and their glucose levels are trending high, it alerts the person to help make diabetes management easier and provides peace of mind," Dr. Sandra Schlüter, Endocrinologist, Germany, and head of AGDF told the press.
By combining Simplera and InPen to form its Smart MDI solution, Medtronic hopes help diabetes patients reach their glycemic goals by offering actionable insights to reduce guesswork and manual calculations.
"Despite the rapid adoption of CGM over the past decade, less than 30% of individuals on MDI therapy using a CGM achieve glycemic targets—highlighting a significant unmet need. We're excited to help more people to reach their goals with our advanced algorithm in InPen powered by our smallest and most comfortable CGM to-date," said Que Dallara, EVP and president of Medtronic Diabetes. "This newest addition of a Smart MDI solution to our holistic portfolio demonstrates our commitment to meeting people where they are in their diabetes journey with simplified solutions that help make life with diabetes easier."
Disclaimer: Simplera is indicated for patients aged two and over and is compatible with iOS and Android operating systems. It’s not yet approved by the U.S. Food and Drug Administration (FDA) and limited to investigational use here.
Simplera touts Medtronic’s newest, no-fingerstick sensor (which requires no over tape) and integrates with InPen—the company’s smart insulin pen that leverages an adjustable algorithm to add real-time, personalized dosing guidance for diabetes management.
"Patients with diabetes can get overwhelmed by the sheer number of decisions they need to make on a daily basis. As a physician, I appreciate the ability to introduce this solution by Medtronic as it provides real-time, personalized guidance to help patients stay in range. For instance, when it detects someone is consuming a meal and their glucose levels are trending high, it alerts the person to help make diabetes management easier and provides peace of mind," Dr. Sandra Schlüter, Endocrinologist, Germany, and head of AGDF told the press.
By combining Simplera and InPen to form its Smart MDI solution, Medtronic hopes help diabetes patients reach their glycemic goals by offering actionable insights to reduce guesswork and manual calculations.
"Despite the rapid adoption of CGM over the past decade, less than 30% of individuals on MDI therapy using a CGM achieve glycemic targets—highlighting a significant unmet need. We're excited to help more people to reach their goals with our advanced algorithm in InPen powered by our smallest and most comfortable CGM to-date," said Que Dallara, EVP and president of Medtronic Diabetes. "This newest addition of a Smart MDI solution to our holistic portfolio demonstrates our commitment to meeting people where they are in their diabetes journey with simplified solutions that help make life with diabetes easier."
Disclaimer: Simplera is indicated for patients aged two and over and is compatible with iOS and Android operating systems. It’s not yet approved by the U.S. Food and Drug Administration (FDA) and limited to investigational use here.