Sam Brusco, Associate Editor09.15.23
Attune Medical was awarded De Novo authorization from the U.S. Food and Drug Administration (FDA) for its ensoETM device to reduce likelihood of ablation-related esophageal injury caused by radiofrequency (RF) cardiac ablation procedures.
Published studies that assessed use of ensoETM for this purpose include the pilot eCOOL-AF study, the IMPACT study and a large multi-center analysis of over 25,000 patients (in press at the Journal of the American College of Cardiology: Clinical Electrophysiology).
The studies found up to an 83% esophageal injury reduction and “significant reduction in risk” of atrioesphageal fistula (AEF), one of the most dangerous complications of ablation procedures.
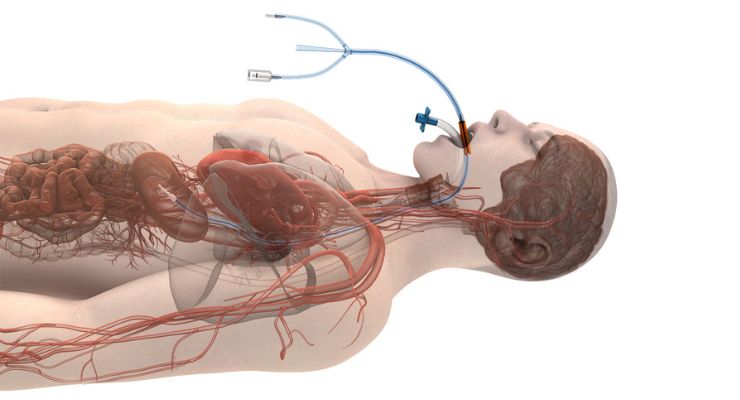
ensoETM is a single-use, thermal regulating device placed in the esophagus—similar to an orogastric tube—and connected to an external heat exchange unit, making a closed-loop system for proactive, controlled temperature management. It’s been cleared to control patient temperature since 2015, and Attune Medical said over 50,000 patients to date have been treated in critical care units, emergency rooms, operating rooms, and electrophysiology labs.
“Historically, there have been no proven strategies to prevent esophageal injury during ablation procedures, and injury rates have not declined despite the use of temperature probes,” Jason Zagrodzky, MD, FHRS, Electrophysiologist at Texas Cardiac Arrhythmia in Austin, Texas told the press. “This De Novo authorization gives electrophysiologists a solution to proactively cool the esophageal wall during ablation procedures and is a great leap forward in best practice standards and patient care. We have found a 35 percent reduction in fluoroscopy requirements with cooling, as well as a reduced staff workload which results in significant cost savings to hospital systems.”
“Our lab has published studies showing a 30 percent reduction in procedure time and a 14 percent improvement in long-term efficacy when using ensoETM,” noted Mark Metzl, MD, FHRS, Section Chief, Cardiac Electrophysiology at NorthShore University Health System. “These findings have in turn been shown to result in a quicker return home for patients and a cost savings of up to $2,135 per procedure.”
“Over the last 20 years, significant resources have been committed to mitigating serious esophageal complications, with no meaningful results. This De Novo marketing authorization opens the door to a new standard. Studies have shown improved safety and efficacy for patients while allowing improved efficiency for physicians and greater cost savings for the hospitals in which they operate,” added Jay Istvan, Attune Medical’s CEO.
Published studies that assessed use of ensoETM for this purpose include the pilot eCOOL-AF study, the IMPACT study and a large multi-center analysis of over 25,000 patients (in press at the Journal of the American College of Cardiology: Clinical Electrophysiology).
The studies found up to an 83% esophageal injury reduction and “significant reduction in risk” of atrioesphageal fistula (AEF), one of the most dangerous complications of ablation procedures.
ensoETM is a single-use, thermal regulating device placed in the esophagus—similar to an orogastric tube—and connected to an external heat exchange unit, making a closed-loop system for proactive, controlled temperature management. It’s been cleared to control patient temperature since 2015, and Attune Medical said over 50,000 patients to date have been treated in critical care units, emergency rooms, operating rooms, and electrophysiology labs.
“Historically, there have been no proven strategies to prevent esophageal injury during ablation procedures, and injury rates have not declined despite the use of temperature probes,” Jason Zagrodzky, MD, FHRS, Electrophysiologist at Texas Cardiac Arrhythmia in Austin, Texas told the press. “This De Novo authorization gives electrophysiologists a solution to proactively cool the esophageal wall during ablation procedures and is a great leap forward in best practice standards and patient care. We have found a 35 percent reduction in fluoroscopy requirements with cooling, as well as a reduced staff workload which results in significant cost savings to hospital systems.”
“Our lab has published studies showing a 30 percent reduction in procedure time and a 14 percent improvement in long-term efficacy when using ensoETM,” noted Mark Metzl, MD, FHRS, Section Chief, Cardiac Electrophysiology at NorthShore University Health System. “These findings have in turn been shown to result in a quicker return home for patients and a cost savings of up to $2,135 per procedure.”
“Over the last 20 years, significant resources have been committed to mitigating serious esophageal complications, with no meaningful results. This De Novo marketing authorization opens the door to a new standard. Studies have shown improved safety and efficacy for patients while allowing improved efficiency for physicians and greater cost savings for the hospitals in which they operate,” added Jay Istvan, Attune Medical’s CEO.