Michael Barbella, Managing Editor09.13.23
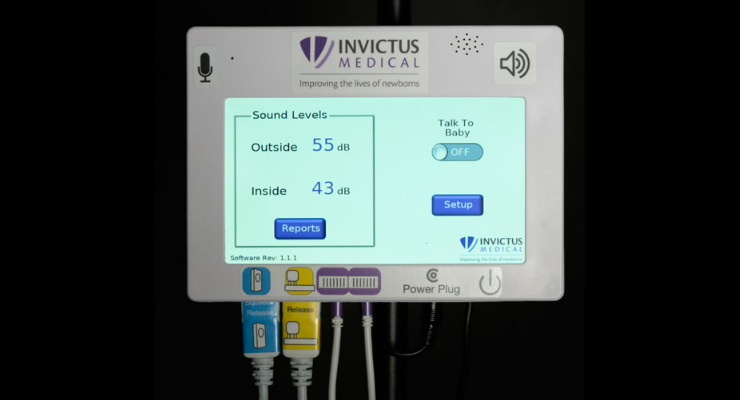
Invictus Medical has received U.S. Food and Drug Administration clearance for its Neoasis incubator-based active noise control (ANC) device.
“With this clearance for use, Invictus has made a huge step towards deploying the Neoasis ANC device in neonatal intensive care units. It is well documented that a quieter environment has a positive impact, including improved sleep hygiene and weight gain in infants where both are critical for development,” Invictus Medical CEO George Hutchinson, Ph.D., said.
The Neoasis utilizes a proprietary, innovative active noise control (ANC) system to attenuate noise with canceling sound wave technology. At the same time, it allows a parent’s voice to be directed to the infant, which can also be beneficial for cognitive development.
“The increasing sophistication of NICU care has created a burden of noxious stimuli for critically ill infants,” said Dr. Robert White, practice medical director, Pediatrix Medical Group. “The rapidly growing auditory cortex of these neonates can only develop properly when background noise doesn't interfere with nurturing sounds of parents and caregivers.”
The functionality and performance of the Neoasis were highlighted in a presentation this past summer at the Pediatric Academic Society conference. “In our study, we sought to measure the level of noise reduction provided by commercially available neonatal ear covers in a simulated NICU environment and compare this to levels provided by this novel noise reduction device designed specifically for use in neonatal incubators,” Hutchinson explained.
“What was demonstrated and what our article shows is that the Neoasis ANC device is effective over a broad area in the incubator and is superior to commercially available adhesive ear muffs. In previously published clinical studies, this level of noise attenuation resulted in improved sleep hygiene and rate of growth.”
Invictus Medical is a San Antonio, Texas-based privately-held company that aims to ensure that children attain their developmental milestones. .
“With this clearance for use, Invictus has made a huge step towards deploying the Neoasis ANC device in neonatal intensive care units. It is well documented that a quieter environment has a positive impact, including improved sleep hygiene and weight gain in infants where both are critical for development,” Invictus Medical CEO George Hutchinson, Ph.D., said.
The Neoasis utilizes a proprietary, innovative active noise control (ANC) system to attenuate noise with canceling sound wave technology. At the same time, it allows a parent’s voice to be directed to the infant, which can also be beneficial for cognitive development.
“The increasing sophistication of NICU care has created a burden of noxious stimuli for critically ill infants,” said Dr. Robert White, practice medical director, Pediatrix Medical Group. “The rapidly growing auditory cortex of these neonates can only develop properly when background noise doesn't interfere with nurturing sounds of parents and caregivers.”
The functionality and performance of the Neoasis were highlighted in a presentation this past summer at the Pediatric Academic Society conference. “In our study, we sought to measure the level of noise reduction provided by commercially available neonatal ear covers in a simulated NICU environment and compare this to levels provided by this novel noise reduction device designed specifically for use in neonatal incubators,” Hutchinson explained.
“What was demonstrated and what our article shows is that the Neoasis ANC device is effective over a broad area in the incubator and is superior to commercially available adhesive ear muffs. In previously published clinical studies, this level of noise attenuation resulted in improved sleep hygiene and rate of growth.”
Invictus Medical is a San Antonio, Texas-based privately-held company that aims to ensure that children attain their developmental milestones. .