Sam Brusco, Associate Editor09.06.23
Techsomed, an Israeli software developer aiming to enhance clinical impact for ablation therapy, has received U.S. Food and Drug Administration (FDA) 510(k) clearance for its VisAble.IO software to help plan liver ablation procedures and confirm ablation zones.
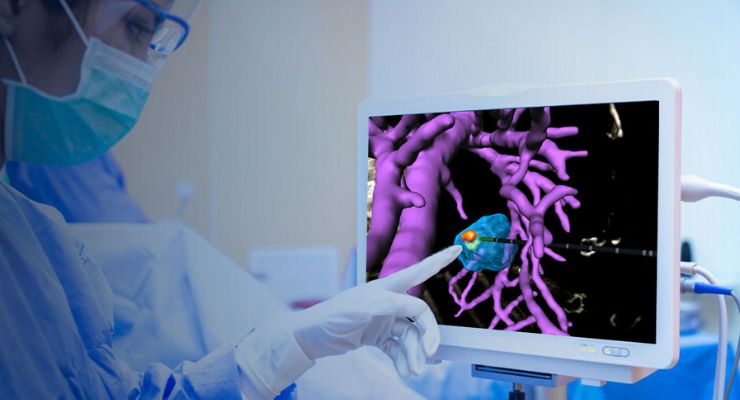
VisAble.IO is the first product to roll out as part of the company’s BioTrace solution, an end-to-end software tool for full visualization and control during image-guided ablation therapy. BioTrace uses AI-based tech to leverage standard imaging like ultrasound, CT, or MRI to offer visualization to the ablation zone’s full extent in real-time—together with smart, personalized treatment planning and assessment tools.
Advanced computation and image registration help to plan and asses a liver tumor’s ablation coverage. 3D visualization of the ablation target in a patient-specific, anatomical view, virtual instrument overly and position, estimated ablation regions, and an interactive, 3D view of ablation margins and missed volume help evaluate ablation target coverage immediately post-procedure.
"We are excited to receive 510(k) clearance for our VisAble.IO solution and appreciate the relentless efforts of our entire team in achieving this milestone. This is a pivotal step forward in our journey of elevating ablation therapy into image guided ablation therapy and making it available for every patient everywhere," Yossi Abu, CEO and founder of Techsomed told the press. "The FDA's validation of VisAble.IO's safety and effectiveness not only reflects the exceptional quality of our product but also paves the way for significant commercial opportunities".
Techsomed’s BioTrace.IO Lite software is currently under FDA review through the De Novo pathway after successfully completing a U.S. pivotal trial. The company said BioTrace.IO Lite will be the first software device to provide visualization of the ablated area based on standard real-time ultrasound imaging.
VisAble.IO is the first product to roll out as part of the company’s BioTrace solution, an end-to-end software tool for full visualization and control during image-guided ablation therapy. BioTrace uses AI-based tech to leverage standard imaging like ultrasound, CT, or MRI to offer visualization to the ablation zone’s full extent in real-time—together with smart, personalized treatment planning and assessment tools.
Advanced computation and image registration help to plan and asses a liver tumor’s ablation coverage. 3D visualization of the ablation target in a patient-specific, anatomical view, virtual instrument overly and position, estimated ablation regions, and an interactive, 3D view of ablation margins and missed volume help evaluate ablation target coverage immediately post-procedure.
"We are excited to receive 510(k) clearance for our VisAble.IO solution and appreciate the relentless efforts of our entire team in achieving this milestone. This is a pivotal step forward in our journey of elevating ablation therapy into image guided ablation therapy and making it available for every patient everywhere," Yossi Abu, CEO and founder of Techsomed told the press. "The FDA's validation of VisAble.IO's safety and effectiveness not only reflects the exceptional quality of our product but also paves the way for significant commercial opportunities".
Techsomed’s BioTrace.IO Lite software is currently under FDA review through the De Novo pathway after successfully completing a U.S. pivotal trial. The company said BioTrace.IO Lite will be the first software device to provide visualization of the ablated area based on standard real-time ultrasound imaging.