Sam Brusco, Associate Editor08.30.23
LivaNova has earned U.S. Food and Drug Administration (FDA) 510(k) clearance and CE mark approval for its Essenz in-line blood monitor (ILBM), which offers continuous, accurate measurement of essential blood parameters throughout cardiopulmonary bypass (CPB) procedures.
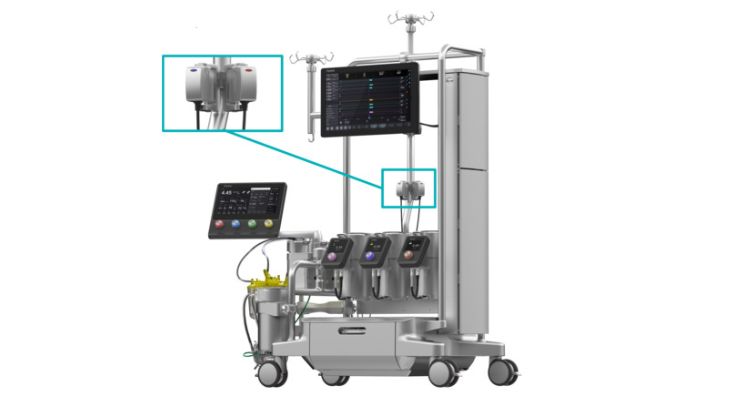
The ILBM is integrated into the company’s next-gen Essenz perfusion system CPB platform, which lets perfusionists access and manage reliable blood parameters from the system’s cockpit without needing further monitors or holders. With Essenz ILBM, perfusionists get in-line continuous monitoring of patient parameters for the procedure’s duration.
This allows delivery of a patient-tailored approach to perfusion, rooted in data-driven decisions.
“Dynamic conditions can rapidly change a patient’s blood parameters during a cardiopulmonary bypass procedure,” Marco Dolci, LivaNova President, Cardiopulmonary told the press. “The Essenz In-Line Blood Monitor provides continuous monitoring throughout a patient’s procedure. Access to accurate, real-time measurements directly from the Essenz Perfusion System allows for quick decisions and tailored care strategies to serve the patient.”
Essenz ILBM is powered by LivaNova’s B-Capta sensing tech, and the company said it’s the only in-line blood monitoring system that works within CLIA guidelines and offers parameter values in line with hospital blood gas analyzers. The ILBM provides measured values for oxygen saturation, hematocrit, partial pressure of oxygen and temperature, instead of calculated values.
It also doesn’t need calibration to set device measurements so time can be saved during setup. Arterial and venous parameters are automatically transferred to the Essenz patient monitor, supporting data-driven decision making and the implementation of goal-directed perfusion (GDP).
The ILBM is integrated into the company’s next-gen Essenz perfusion system CPB platform, which lets perfusionists access and manage reliable blood parameters from the system’s cockpit without needing further monitors or holders. With Essenz ILBM, perfusionists get in-line continuous monitoring of patient parameters for the procedure’s duration.
This allows delivery of a patient-tailored approach to perfusion, rooted in data-driven decisions.
“Dynamic conditions can rapidly change a patient’s blood parameters during a cardiopulmonary bypass procedure,” Marco Dolci, LivaNova President, Cardiopulmonary told the press. “The Essenz In-Line Blood Monitor provides continuous monitoring throughout a patient’s procedure. Access to accurate, real-time measurements directly from the Essenz Perfusion System allows for quick decisions and tailored care strategies to serve the patient.”
Essenz ILBM is powered by LivaNova’s B-Capta sensing tech, and the company said it’s the only in-line blood monitoring system that works within CLIA guidelines and offers parameter values in line with hospital blood gas analyzers. The ILBM provides measured values for oxygen saturation, hematocrit, partial pressure of oxygen and temperature, instead of calculated values.
It also doesn’t need calibration to set device measurements so time can be saved during setup. Arterial and venous parameters are automatically transferred to the Essenz patient monitor, supporting data-driven decision making and the implementation of goal-directed perfusion (GDP).