Sam Brusco, Associate Editor08.22.23
Regenesis Biomedical, a company focused on non-drug pain management, gained U.S. Food and Drug Administration (FDA) clearance and began commercialization of its Reprieve by Regenesis device.
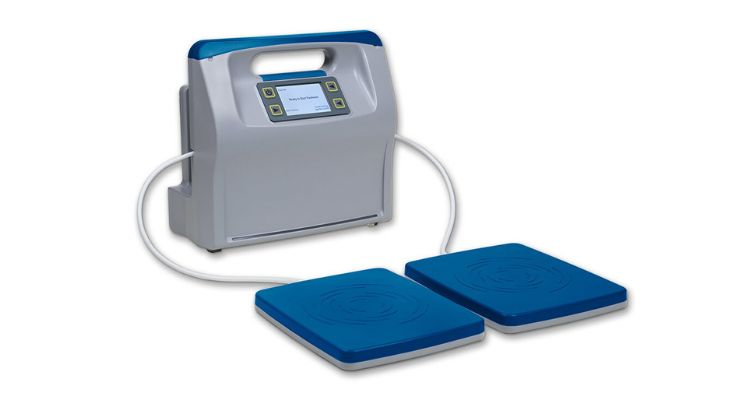
According to the company, Reprieve is the only home-use, shortwave diathermy device on the market. It’s based on Regenesis’ pulsed electromagnetic technology platform.
"In alignment with our Vision to improve health for a longer, more active, and fulfilling life, we are pleased to announce we received [FDA] clearance for our new Reprieve by Regenesis device," Tom Eisiminger, Regenesis president and CEO told the press.
Regenesis researches, designs, manufactures, and sells energy-based products to alleviate pain, restore health, and improve quality of life. Focusing on veterans’ needs via direct salesforce, the company claims over 23,000 patients have been treated with its non-drug, electromagnetic energy to manage pain.
Eisiminger continued, "Reprieve allows us to market our therapy for broad pain indications within the Department of Veterans Health Administration (VA), which represents a market that is 16X the size of our current market. Because our new device has a two-treatment applicator option, we are expanding our efforts into the podiatric space with a focus on the treatment of pain associated with diabetic neuropathy."
According to the company, Reprieve is the only home-use, shortwave diathermy device on the market. It’s based on Regenesis’ pulsed electromagnetic technology platform.
"In alignment with our Vision to improve health for a longer, more active, and fulfilling life, we are pleased to announce we received [FDA] clearance for our new Reprieve by Regenesis device," Tom Eisiminger, Regenesis president and CEO told the press.
Regenesis researches, designs, manufactures, and sells energy-based products to alleviate pain, restore health, and improve quality of life. Focusing on veterans’ needs via direct salesforce, the company claims over 23,000 patients have been treated with its non-drug, electromagnetic energy to manage pain.
Eisiminger continued, "Reprieve allows us to market our therapy for broad pain indications within the Department of Veterans Health Administration (VA), which represents a market that is 16X the size of our current market. Because our new device has a two-treatment applicator option, we are expanding our efforts into the podiatric space with a focus on the treatment of pain associated with diabetic neuropathy."