Michael Barbella, Managing Editor08.22.23
Reflow Medical Inc. is sharing six-month results of the DEEPER OUS clinical trial, evaluating the safety and efficacy of the Bare Temporary Spur Stent System.
Michael Lichtenberg, M.D., chief medical officer and director of the Angiology and Vascular Center in Arnsberg, Germany, said the Bare Temporary Spur Stent System used in conjunction with a commercially available paclitaxel-coated balloon met:
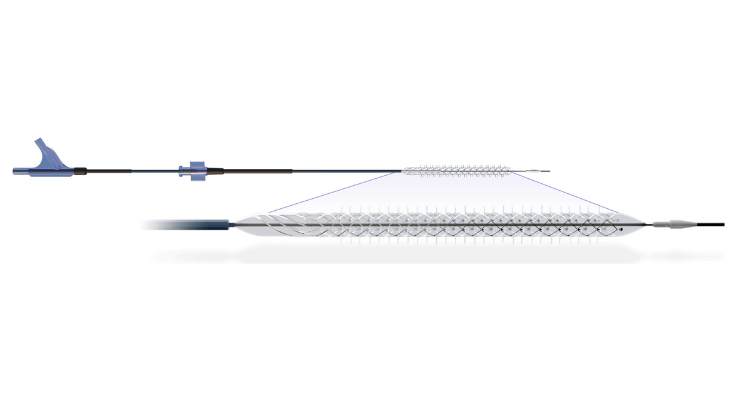
The Bare Temporary Spur Stent System is designed to deliver a temporary stent with integrated radial spikes to the peripheral vasculature via a sheathed delivery system, for stent-like results while leaving nothing behind.
“It’s gratifying to see the improvement in our patients six months after treatment with the Spur Stent System,” Lichtenberg stated. “We observed improvements in their Rutherford class scores, and fewer than 8% required revascularization during the six-month period.”
According to Prof. Thomas Zeller, M.D., chief of the Department of Angiology at University Heart Center Freiburg-Bad Krozingen, the sub-study results also showed that “calcification did not appear to have an effect on vessel patency or vessel recoil, suggesting that the Bare Temporary Spur Stent System may modify calcification, decrease recoil and enhance drug uptake.”
“We’re very grateful to Dr. Lichtenberg, Prof. Zeller, and the other investigators and teams involved in the trial, as well as to the patients for their participation,” Reflow Medical Senior Vice President for Research and Development Teo Jimenez said. “We’re looking forward to continuing to see great results from the study.”
The Bare Temporary Spur Stent System is for investigational use only. Follow-up to five years is ongoing.
Established in 2011, Reflow Medicalis a private company that designs and develops technologies for treating cardiovascular disease.
Michael Lichtenberg, M.D., chief medical officer and director of the Angiology and Vascular Center in Arnsberg, Germany, said the Bare Temporary Spur Stent System used in conjunction with a commercially available paclitaxel-coated balloon met:
- the primary effectiveness endpoint with an 86% primary patency rate at six months, and
- the primary safety endpoint, i.e., freedom from target limb major adverse limb event (MALE) and all-cause perioperative death (POD) at 30 days, in all subjects.
The Bare Temporary Spur Stent System is designed to deliver a temporary stent with integrated radial spikes to the peripheral vasculature via a sheathed delivery system, for stent-like results while leaving nothing behind.
“It’s gratifying to see the improvement in our patients six months after treatment with the Spur Stent System,” Lichtenberg stated. “We observed improvements in their Rutherford class scores, and fewer than 8% required revascularization during the six-month period.”
According to Prof. Thomas Zeller, M.D., chief of the Department of Angiology at University Heart Center Freiburg-Bad Krozingen, the sub-study results also showed that “calcification did not appear to have an effect on vessel patency or vessel recoil, suggesting that the Bare Temporary Spur Stent System may modify calcification, decrease recoil and enhance drug uptake.”
“We’re very grateful to Dr. Lichtenberg, Prof. Zeller, and the other investigators and teams involved in the trial, as well as to the patients for their participation,” Reflow Medical Senior Vice President for Research and Development Teo Jimenez said. “We’re looking forward to continuing to see great results from the study.”
The Bare Temporary Spur Stent System is for investigational use only. Follow-up to five years is ongoing.
Established in 2011, Reflow Medicalis a private company that designs and develops technologies for treating cardiovascular disease.