Sam Brusco, Associate Editor08.22.23
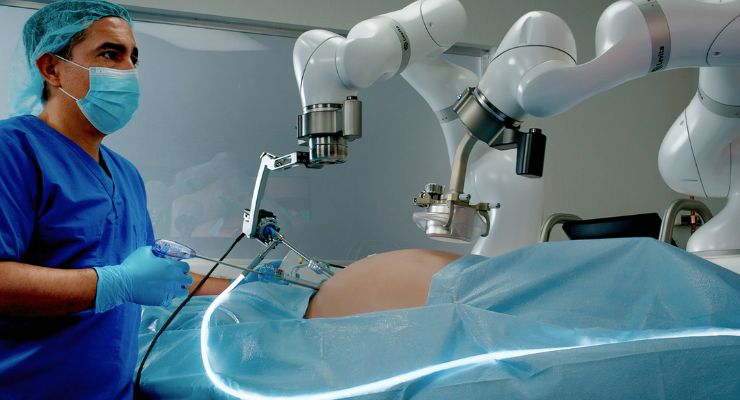
Levita Magnetics has gained U.S. Food and Drug Administration (FDA) clearance for its MARS (Magnetic-Assisted Robotic Surgery) platform, a minimally invasive surgical platform for high-volume abdominal surgery.
The MARS system uses magnets and machines to reduce the number of incisions and give surgeons complete control during laparoscopic procedures, according to Levita. The company said it’s in a compact footprint engineered to fit inside existing operating rooms.
This news follows a $26 million Series C financing round in August 2022 to support regulatory and commercial efforts for MARS.
"Today marks a significant milestone in Levita's mission to provide more patients access to state-of-the-art surgical technology. MARS is poised to revolutionize surgical options for a broad range of patients," Dr. Alberto Rodriguez-Navarro, surgeon, founder, and CEO of Levita Magnetics, told the press. "With this FDA clearance, we eagerly anticipate making a substantial impact across the value chain."
Levita said it designed MARS to offer the same patient benefits as its proprietary Magnetic Surgery. Less-invasive surgery with reduced incisions can bring about less pain, faster recovery, and fewer scars. Full control over the laparoscopic view removes the need for an additional assistant, and full control over the Magnetic Surgical system allows retraction of large tissues/organs like the liver, gallbladder, prostate, and colon.
"MARS by Levita has the potential to reshape the surgical industry and forever change medical innovation. Our pioneering MARS platform gives patients and surgeons a transformative tool that will usher in a fundamental shift in surgery for years to come," said Levita chairperson Maria Sainz. "Levita's system can aid in surgeon proficiency and efficiency, and can reduce the need for assistive personnel, signaling a major achievement not only for Levita but for surgical care."
MARS builds on the success of the first generation Levita Magnetic Surgical system, which initially earned FDA clearance for gallbladder surgery in 2016. An expanded indication was granted from the agency for bariatric procedures in 2018, and another for prostatectomies was awarded in 2019.
The MARS system uses magnets and machines to reduce the number of incisions and give surgeons complete control during laparoscopic procedures, according to Levita. The company said it’s in a compact footprint engineered to fit inside existing operating rooms.
This news follows a $26 million Series C financing round in August 2022 to support regulatory and commercial efforts for MARS.
"Today marks a significant milestone in Levita's mission to provide more patients access to state-of-the-art surgical technology. MARS is poised to revolutionize surgical options for a broad range of patients," Dr. Alberto Rodriguez-Navarro, surgeon, founder, and CEO of Levita Magnetics, told the press. "With this FDA clearance, we eagerly anticipate making a substantial impact across the value chain."
Levita said it designed MARS to offer the same patient benefits as its proprietary Magnetic Surgery. Less-invasive surgery with reduced incisions can bring about less pain, faster recovery, and fewer scars. Full control over the laparoscopic view removes the need for an additional assistant, and full control over the Magnetic Surgical system allows retraction of large tissues/organs like the liver, gallbladder, prostate, and colon.
"MARS by Levita has the potential to reshape the surgical industry and forever change medical innovation. Our pioneering MARS platform gives patients and surgeons a transformative tool that will usher in a fundamental shift in surgery for years to come," said Levita chairperson Maria Sainz. "Levita's system can aid in surgeon proficiency and efficiency, and can reduce the need for assistive personnel, signaling a major achievement not only for Levita but for surgical care."
MARS builds on the success of the first generation Levita Magnetic Surgical system, which initially earned FDA clearance for gallbladder surgery in 2016. An expanded indication was granted from the agency for bariatric procedures in 2018, and another for prostatectomies was awarded in 2019.