Sam Brusco, Associate Editor07.12.23
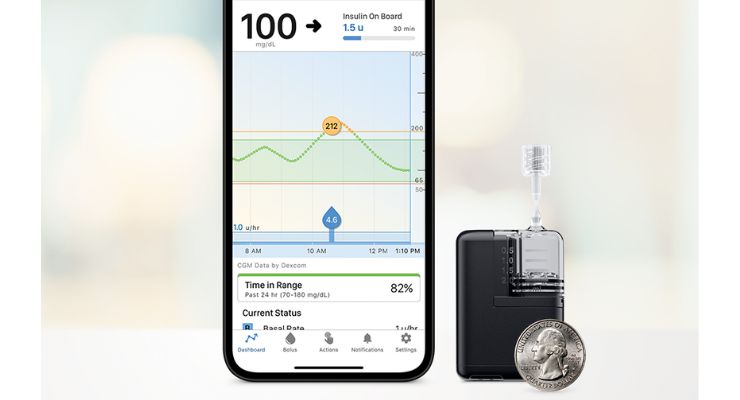
Tandem Diabetes Care has gained U.S. Food and Drug Administration (FDA) clearance for the Tandem Mobi insulin pump for patients with diabetes aged six and up. Tandem Mobi, according to the company, is fully controllable via a mobile app and is the smallest durable automated insulin delivery (AID) system on the market.
“Testing the limits of pump miniaturization, Tandem Mobi joins the t:slim X2 pump in our family of insulin delivery solutions bringing new options in wearability, the flexibility to disconnect, and full phone control,” John Sheridan, Tandem Diabetes’ president and CEO told the press. “Through this expansion, we are delivering on our commitment to bring greater choice, along with the proven benefits of Tandem’s technology, to more people living with diabetes.”
Tandem Mobi touts a 200-unit insulin cartridge and on-pump button as an alternative option to phone control to dose insulin. Less than half the size of the t:slim X2 pump, Mobi can fit in a coin pocket, clipped to clothing, or worn on the body with an adhesive sleeve.
Mobi is also compatible with all Tandem-branded infusion sets made by the Convatec Group, including a new, five-inch tubing option made for Mobi. It also features Tandem’s Control-IQ technology, a hybrid, closed-loop algorithm to predict glucose levels 30 minutes ahead and adjust insulin delivery every five minutes.
Full iOS mobile control is enabled through the user’s iPhone. The AID system also features inductive charging and is capable of wireless remote software updates using a compatible smartphone.
Tandem expects to begin a limited release of Mobi in late 2023, with full commercial launch aimed for early 2024.
“Testing the limits of pump miniaturization, Tandem Mobi joins the t:slim X2 pump in our family of insulin delivery solutions bringing new options in wearability, the flexibility to disconnect, and full phone control,” John Sheridan, Tandem Diabetes’ president and CEO told the press. “Through this expansion, we are delivering on our commitment to bring greater choice, along with the proven benefits of Tandem’s technology, to more people living with diabetes.”
Tandem Mobi touts a 200-unit insulin cartridge and on-pump button as an alternative option to phone control to dose insulin. Less than half the size of the t:slim X2 pump, Mobi can fit in a coin pocket, clipped to clothing, or worn on the body with an adhesive sleeve.
Mobi is also compatible with all Tandem-branded infusion sets made by the Convatec Group, including a new, five-inch tubing option made for Mobi. It also features Tandem’s Control-IQ technology, a hybrid, closed-loop algorithm to predict glucose levels 30 minutes ahead and adjust insulin delivery every five minutes.
Full iOS mobile control is enabled through the user’s iPhone. The AID system also features inductive charging and is capable of wireless remote software updates using a compatible smartphone.
Tandem expects to begin a limited release of Mobi in late 2023, with full commercial launch aimed for early 2024.