Sam Brusco, Associate Editor06.26.23
Medtronic presented a collection of new clinical and real-world data on its MiniMed 780G system at the 83rd American Diabetes Association (ADA) Scientific Sessions.
The data sets evaluated a wide range of users, including historically challenging younger patients, those not meeting blood sugar goals, and those using a simplified meal announcement leveraging fixed carbohydrate amount instead of exact carb calculations. The data sets found proprietary Meal Detection tech supported Time in Range (TIR) outcomes that surpass consensus guidelines of 70%.
Further, the system is helping lower the percent of time for both children and adults spent in hyperglycemia.
Results showed those using the fixed carb entry kept international glycemic control targets, including A1c of 6.9% and TIR of 72.7% vs. 79.4% in the precise entry group over six months. Further, the fixed entry group lowered time above 250 mg/dL from 28.3% to 5.3% at six months vs. 3.9% in the precise entry group. After three months, 88% chose to continue with fixed meal management, suggesting user satisfaction with the less burdensome approach.
"Many individuals with type 1 diabetes struggle with meal management with nearly 50% considering carb counting the most burdensome aspect of diabetes management. Indeed, many frequently underestimate their carbs or forget to bolus and this has an adverse impact on clinical outcomes," Dr. Petrovski told the press. "This study shows that a simplified meal management approach with the MiniMed 780G system helped users maintain glycemic targets while providing forgiveness for inexact carb counts. Clearly there's more runway for simplification of diabetes management with this system and it's promising for the many patients struggling with meal management."
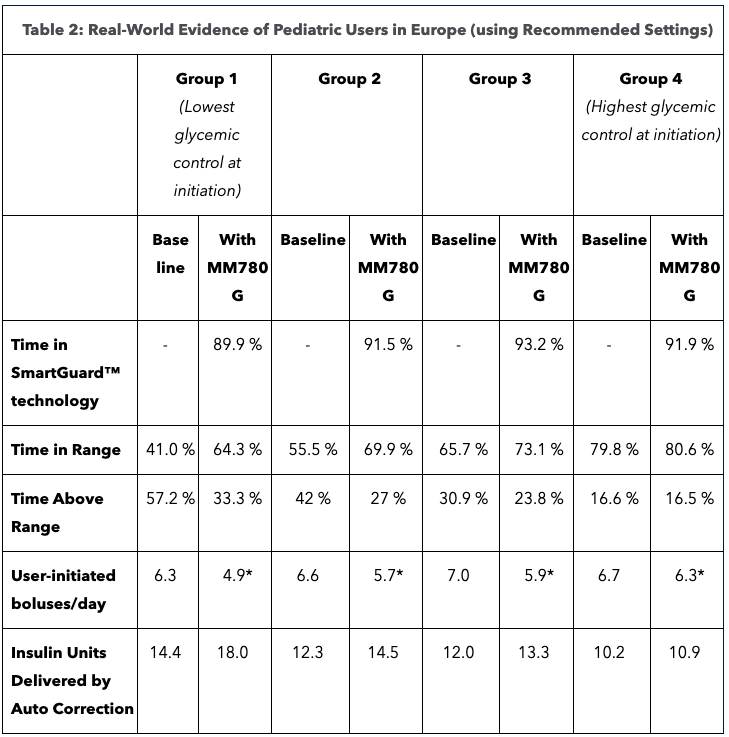
The group with the lowest TIR before SmartGuard has the largest increase of 23.3% in TIR. Patients with the best metabolic control in SmartGuard achieved 80.6% TIR. The increase in TIR was seen with less effort, evidence by fewer user-initiated boluses.
Real-world data from 108 patients on the Medtronic Extended Infusion set was presented for the first time as well, and analysis demonstrated 6.74-day average infusion set wear time. 48.2% of the individuals evaluated wore the set for seven days, mirroring results from the U.S. pivotal trial and delivering less user burden through less frequent infusion set changes.
"We're committed to pushing simplification of diabetes management as far as we can and are heartened to see the impact our MiniMed 780G system is having on both clinical and quality of life outcomes as evidenced by our randomized controlled ADAPT study and the growing body of real-world evidence from around the world," said Que Dallara, EVP and president of Medtronic Diabetes. "With each advancement, we're working to reduce more of the burden that this disease demands and will continue to innovate to make life easier for those we have the privilege to support."
The data sets evaluated a wide range of users, including historically challenging younger patients, those not meeting blood sugar goals, and those using a simplified meal announcement leveraging fixed carbohydrate amount instead of exact carb calculations. The data sets found proprietary Meal Detection tech supported Time in Range (TIR) outcomes that surpass consensus guidelines of 70%.
Further, the system is helping lower the percent of time for both children and adults spent in hyperglycemia.
Simplified (Fixed) Meal Announcements with MiniMed 780G
The first study by Dr. Goran Petrovski, MD, Ph.D., of Sidra Medicine, randomly assigned 34 adolescents using MiniMed 780G into two groups, with some entering a fixed pre-set number of carbs (small, average, or high) and some calculating a precise number of carbs for meals. The subjects lived with diabetes for at least a year and used multiple daily injections or pump therapy before the study.Results showed those using the fixed carb entry kept international glycemic control targets, including A1c of 6.9% and TIR of 72.7% vs. 79.4% in the precise entry group over six months. Further, the fixed entry group lowered time above 250 mg/dL from 28.3% to 5.3% at six months vs. 3.9% in the precise entry group. After three months, 88% chose to continue with fixed meal management, suggesting user satisfaction with the less burdensome approach.
"Many individuals with type 1 diabetes struggle with meal management with nearly 50% considering carb counting the most burdensome aspect of diabetes management. Indeed, many frequently underestimate their carbs or forget to bolus and this has an adverse impact on clinical outcomes," Dr. Petrovski told the press. "This study shows that a simplified meal management approach with the MiniMed 780G system helped users maintain glycemic targets while providing forgiveness for inexact carb counts. Clearly there's more runway for simplification of diabetes management with this system and it's promising for the many patients struggling with meal management."
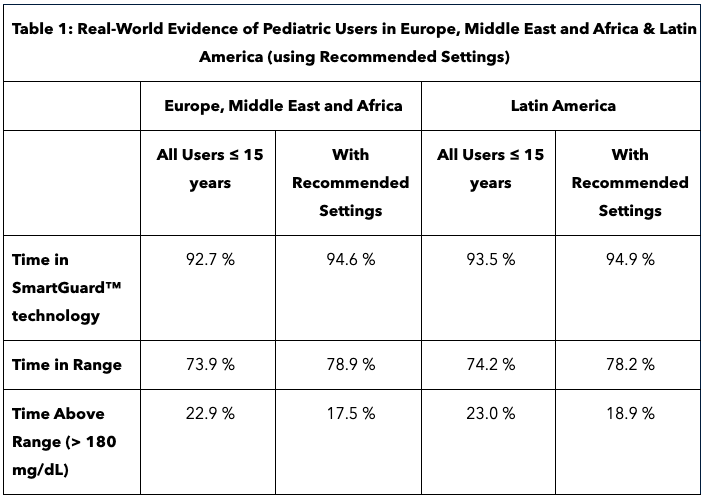
Real-World Performance in Children
Analysis of real-world evidence of 3,543 children 15 and under in Europe and Latin America using MiniMed 780G with recommending settings of 100 mg/dL and two-house active insulin time (AIT) showed TIR of 78%. A separate analysis of 2,516 children 15 and under in Europe also showed improved glycemic performance in SmartGuard tech, regardless of baseline glycemic control.The group with the lowest TIR before SmartGuard has the largest increase of 23.3% in TIR. Patients with the best metabolic control in SmartGuard achieved 80.6% TIR. The increase in TIR was seen with less effort, evidence by fewer user-initiated boluses.


Extended Infusion Set Real-World Data
Real-world data from 108 patients on the Medtronic Extended Infusion set was presented for the first time as well, and analysis demonstrated 6.74-day average infusion set wear time. 48.2% of the individuals evaluated wore the set for seven days, mirroring results from the U.S. pivotal trial and delivering less user burden through less frequent infusion set changes."We're committed to pushing simplification of diabetes management as far as we can and are heartened to see the impact our MiniMed 780G system is having on both clinical and quality of life outcomes as evidenced by our randomized controlled ADAPT study and the growing body of real-world evidence from around the world," said Que Dallara, EVP and president of Medtronic Diabetes. "With each advancement, we're working to reduce more of the burden that this disease demands and will continue to innovate to make life easier for those we have the privilege to support."












