Michael Barbella, Managing Editor06.26.23
Lumicell Inc. is sharing promising results of the Investigation of Novel Surgical Imaging for Tumor Excision (INSITE) pivotal trial. Data prove the safety and effectiveness of the company’s investigational Lumicell Direct Visualization System (DVS) in identifying residual breast cancer after removal of the primary specimen during initial lumpectomy.
In the INSITE trial, the Lumicell DVS identified cancer remaining after the standard of care lumpectomy procedure in approximately 8% of patients. In the majority of these patients, standard pathology assessment had deemed margins negative and would have left this tumor behind. The additional residual cancer identified by the Lumicell DVS measured 1 mm to13 mm and included areas of low- and high-grade tumor in patients across all tumor types. In addition, 14.5% of patients with positive margins were converted to final negative margins, potentially avoiding a second surgery. The study also evaluated the safety of Lumicell’s investigational optical imaging agent LUMISIGHT (pegulicianine), with a serious adverse event rate of 0.5%, similar to that of other contrast agents.
“The results of the INSITE trial suggest that a more complete breast cancer resection may be achieved with the Lumicell DVS,” said Barbara Smith, M.D., Ph.D., director of the Breast Program at Massachusetts General Hospital, professor of surgery at Harvard Medical School, and lead investigator of the INSITE clinical trial. “Given the low complication rate, minimal added operative time and, most importantly, the discovery of additional cancer left behind after a lumpectomy, the Lumicell DVS has the potential to be a critical adjunct to enhance standard practice for breast cancer patients.”
The standard of care for lumpectomy surgery involves breast surgeons and pathologists examining the margins of excised tissue to infer whether any residual cancer remains in the breast cavity. Until now, surgeons have had no way to look inside the lumpectomy cavity to find and remove residual cancer during the initial lumpectomy, making it challenging to consistently achieve a complete cancer resection. As a result, 20-40% of lumpectomies have positive margins only identified days after surgery, necessitating a second surgery to obtain clear margins.1,2
“The Lumicell DVS is the first technology designed to enable intracavity examination during the initial lumpectomy surgery, and results of the INSITE study provide evidence of its ability to enable removal of additional residual cancer,” said Jorge Ferrer, Ph.D., senior vice president, clinical research and strategy at Lumicell, and co-inventor of the technology used in the Lumicell DVS. “We believe that publication of this study in a medical journal like NEJM Evidence is a testament to the quality of the research and potential of the Lumicell DVS to improve care and outcomes for breast cancer patients.”
The U.S. Food and Drug Administration (FDA) granted LUMISIGHT Fast Track designation and the Lumicell DVS Breakthrough Device designation in recognition of the potential important benefit of identifying residual cancer during the initial lumpectomy. The New Drug Application (NDA) for LUMISIGHT was submitted to the FDA in March this year and the Premarket Application (PMA) for the Lumicell DVS was submitted to the FDA in April.
The Investigation of Novel Surgical Imaging for Tumor Excision (INSITE) pivotal/Phase 3 trial enrolled 406 patients with primary invasive breast cancer and/or ductal carcinoma in situ (DCIS) across 14 U.S. hospitals. In 27 of 357 patients randomized to the treatment arm (7.6%; 95% CI [5.0%, 10.8%]), Lumicell DVS-guided margins removed tumor left behind after standard lumpectomy, 22 from cavity orientations deemed negative on standard margin evaluation. Conversion from initial positive margins to final negative margins occurred in nine of 62 (14.5%) patients with positive margins by removing a Lumicell DVS-guided margin, potentially avoiding a second surgery in these patients. On per-margin analysis of residual cancer, Lumicell DVS specificity was 85.2% (95% CI [83.7%, 86.6%]) and sensitivity 49.3% (95% CI [37.0%, 61.6%]). LUMISIGHT was generally well-tolerated with a serious adverse event rate of 0.5%, similar to that of other contrast agents.
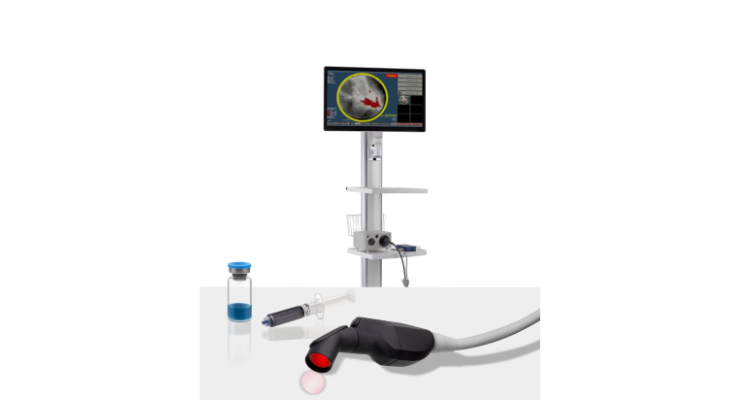
The Lumicell DVS is an investigational system designed for use in patients with breast cancer to assist in the detection of residual malignant tissue within the lumpectomy cavity following removal of the primary specimen during breast conserving surgery. The Lumicell DVS is intended for use with LUMISIGHT (pegulicianine) for fluorescence imaging of the lumpectomy cavity. The investigational Lumicell Direct Visualization System (DVS) is intended for use with the investigational LUMISIGHT Optical Imaging Agent and features a hand-held imaging probe that is designed to go inside the breast cavity and a patient-calibrated cancer detection software to assist in the detection of residual cancer, thereby enabling a more complete resection.
The Lumicell DVS and LUMISIGHT are limited by U.S. law to investigational use only. The Lumicell DVS and LUMISIGHT are not commercially available.
Lumicell is a privately held company using its fluorescence-guided surgical technologies to enable a more complete resection of cancer that may have otherwise been left behind. Lumicell’s proprietary, pan-oncologic optical imaging agent LUMISIGHT is being explored across a wide variety of solid tumor indications.
References
1 Wilke L, Czechura T, Wang C, et al. Repeat surgery after breast conservation for the treatment of stage 0 to II breast carcinoma: a report from the National Cancer Data Base, 2004-2010. JAMA Surg. 2014;149(12):1296-1305.
2 McCahill L, Single R, Aiello Bowles E. Variability in Reexcision Following Breast Conservation Surgery. JAMA. 2012;307(5):467-475.
In the INSITE trial, the Lumicell DVS identified cancer remaining after the standard of care lumpectomy procedure in approximately 8% of patients. In the majority of these patients, standard pathology assessment had deemed margins negative and would have left this tumor behind. The additional residual cancer identified by the Lumicell DVS measured 1 mm to13 mm and included areas of low- and high-grade tumor in patients across all tumor types. In addition, 14.5% of patients with positive margins were converted to final negative margins, potentially avoiding a second surgery. The study also evaluated the safety of Lumicell’s investigational optical imaging agent LUMISIGHT (pegulicianine), with a serious adverse event rate of 0.5%, similar to that of other contrast agents.
“The results of the INSITE trial suggest that a more complete breast cancer resection may be achieved with the Lumicell DVS,” said Barbara Smith, M.D., Ph.D., director of the Breast Program at Massachusetts General Hospital, professor of surgery at Harvard Medical School, and lead investigator of the INSITE clinical trial. “Given the low complication rate, minimal added operative time and, most importantly, the discovery of additional cancer left behind after a lumpectomy, the Lumicell DVS has the potential to be a critical adjunct to enhance standard practice for breast cancer patients.”
The standard of care for lumpectomy surgery involves breast surgeons and pathologists examining the margins of excised tissue to infer whether any residual cancer remains in the breast cavity. Until now, surgeons have had no way to look inside the lumpectomy cavity to find and remove residual cancer during the initial lumpectomy, making it challenging to consistently achieve a complete cancer resection. As a result, 20-40% of lumpectomies have positive margins only identified days after surgery, necessitating a second surgery to obtain clear margins.1,2
“The Lumicell DVS is the first technology designed to enable intracavity examination during the initial lumpectomy surgery, and results of the INSITE study provide evidence of its ability to enable removal of additional residual cancer,” said Jorge Ferrer, Ph.D., senior vice president, clinical research and strategy at Lumicell, and co-inventor of the technology used in the Lumicell DVS. “We believe that publication of this study in a medical journal like NEJM Evidence is a testament to the quality of the research and potential of the Lumicell DVS to improve care and outcomes for breast cancer patients.”
The U.S. Food and Drug Administration (FDA) granted LUMISIGHT Fast Track designation and the Lumicell DVS Breakthrough Device designation in recognition of the potential important benefit of identifying residual cancer during the initial lumpectomy. The New Drug Application (NDA) for LUMISIGHT was submitted to the FDA in March this year and the Premarket Application (PMA) for the Lumicell DVS was submitted to the FDA in April.
The Investigation of Novel Surgical Imaging for Tumor Excision (INSITE) pivotal/Phase 3 trial enrolled 406 patients with primary invasive breast cancer and/or ductal carcinoma in situ (DCIS) across 14 U.S. hospitals. In 27 of 357 patients randomized to the treatment arm (7.6%; 95% CI [5.0%, 10.8%]), Lumicell DVS-guided margins removed tumor left behind after standard lumpectomy, 22 from cavity orientations deemed negative on standard margin evaluation. Conversion from initial positive margins to final negative margins occurred in nine of 62 (14.5%) patients with positive margins by removing a Lumicell DVS-guided margin, potentially avoiding a second surgery in these patients. On per-margin analysis of residual cancer, Lumicell DVS specificity was 85.2% (95% CI [83.7%, 86.6%]) and sensitivity 49.3% (95% CI [37.0%, 61.6%]). LUMISIGHT was generally well-tolerated with a serious adverse event rate of 0.5%, similar to that of other contrast agents.
The Lumicell DVS is an investigational system designed for use in patients with breast cancer to assist in the detection of residual malignant tissue within the lumpectomy cavity following removal of the primary specimen during breast conserving surgery. The Lumicell DVS is intended for use with LUMISIGHT (pegulicianine) for fluorescence imaging of the lumpectomy cavity. The investigational Lumicell Direct Visualization System (DVS) is intended for use with the investigational LUMISIGHT Optical Imaging Agent and features a hand-held imaging probe that is designed to go inside the breast cavity and a patient-calibrated cancer detection software to assist in the detection of residual cancer, thereby enabling a more complete resection.
The Lumicell DVS and LUMISIGHT are limited by U.S. law to investigational use only. The Lumicell DVS and LUMISIGHT are not commercially available.
Lumicell is a privately held company using its fluorescence-guided surgical technologies to enable a more complete resection of cancer that may have otherwise been left behind. Lumicell’s proprietary, pan-oncologic optical imaging agent LUMISIGHT is being explored across a wide variety of solid tumor indications.
References
1 Wilke L, Czechura T, Wang C, et al. Repeat surgery after breast conservation for the treatment of stage 0 to II breast carcinoma: a report from the National Cancer Data Base, 2004-2010. JAMA Surg. 2014;149(12):1296-1305.
2 McCahill L, Single R, Aiello Bowles E. Variability in Reexcision Following Breast Conservation Surgery. JAMA. 2012;307(5):467-475.