Michael Barbella, Managing Editor06.23.23
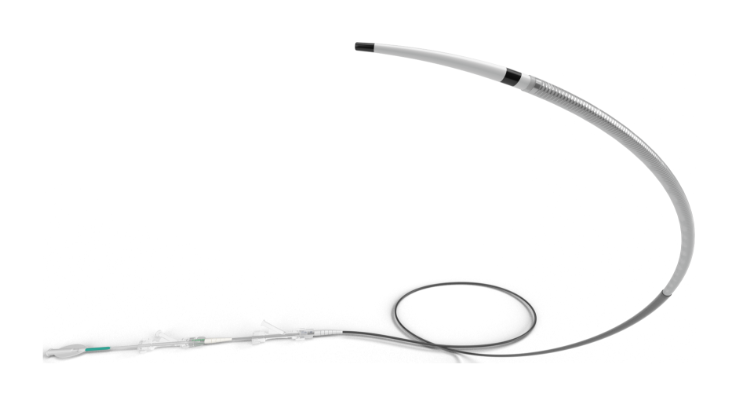
Route 92 Medical Inc. has received U.S. Food and Drug Administration 510(k) clearance for its FreeClimb 70 Reperfusion System, which features the FreeClimb 70 Aspiration Catheter and a Tenzing 7 Delivery Catheter.
The FreeClimb 70 Reperfusion System enables physicians to treat patients experiencing an acute ischemic stroke by removing clots rapidly and safely. This first-to-market system is designed to work together harmoniously for superior deliverability and high procedural efficiency, making it a more refined bi-axial approach for restoring blood flow to the brain during endovascular thrombectomy procedures.
“In our initial experience with FreeClimb 70 and Tenzing 7, the system easily delivered to the target occlusion allowing rapid, effective and safe reperfusion,” said James Caldwell, M.D., an early user of the system at Auckland City Hospital in Auckland, New Zealand. “Tenzing-based delivery can reduce procedure time and may reduce complications. This solution will help clinicians overcome challenging neurovascular anatomy with greater ease and efficiency, providing better care for their patients.”
The FreeClimb 70 Reperfusion System is the first fully integrated solution designed around the Tenzing 7 delivery catheter, which offers superior navigation and predictable access to occluded distal vessels without the need for a guidewire. The system's advanced design eliminates the ledge effect commonly found in large-bore catheters.
"Route 92 Medical aims to provide neurovascular interventionists with innovative endovascular thrombectomy solutions, helping them maximize their opportunity to restore blood flow to the brain on their first attempt,” Route 92 Medical CEO Tony Chou said. "The 510(k) clearance for the FreeClimb 70 Reperfusion System is a key addition to our portfolio of products that are designed to simplify removal of the embolus and seek to improve outcomes.”
The FreeClimb 70 Reperfusion System is part of Route 92 Medical’s catalog of endovascular interventional devices that aim to refine and stroke treatment. The company is currently recruiting participants for the SUMMIT MAX clinical trial, which will support regulatory submissions for the next-generation Monopoint Platform, a super-bore HiPoint 88 catheter delivered with Tenzing 8 for thrombectomy procedures.
Route 92 Medical intends to improve outcomes for patients undergoing neurovascular intervention through cutting-edge engineering and innovative product design. Founded by physicians, the company collaborates with neurovascular clinicians to solve the biggest challenges in neurointervention and deliver differentiated solutions.
The FreeClimb 70 Reperfusion System enables physicians to treat patients experiencing an acute ischemic stroke by removing clots rapidly and safely. This first-to-market system is designed to work together harmoniously for superior deliverability and high procedural efficiency, making it a more refined bi-axial approach for restoring blood flow to the brain during endovascular thrombectomy procedures.
“In our initial experience with FreeClimb 70 and Tenzing 7, the system easily delivered to the target occlusion allowing rapid, effective and safe reperfusion,” said James Caldwell, M.D., an early user of the system at Auckland City Hospital in Auckland, New Zealand. “Tenzing-based delivery can reduce procedure time and may reduce complications. This solution will help clinicians overcome challenging neurovascular anatomy with greater ease and efficiency, providing better care for their patients.”
The FreeClimb 70 Reperfusion System is the first fully integrated solution designed around the Tenzing 7 delivery catheter, which offers superior navigation and predictable access to occluded distal vessels without the need for a guidewire. The system's advanced design eliminates the ledge effect commonly found in large-bore catheters.
"Route 92 Medical aims to provide neurovascular interventionists with innovative endovascular thrombectomy solutions, helping them maximize their opportunity to restore blood flow to the brain on their first attempt,” Route 92 Medical CEO Tony Chou said. "The 510(k) clearance for the FreeClimb 70 Reperfusion System is a key addition to our portfolio of products that are designed to simplify removal of the embolus and seek to improve outcomes.”
The FreeClimb 70 Reperfusion System is part of Route 92 Medical’s catalog of endovascular interventional devices that aim to refine and stroke treatment. The company is currently recruiting participants for the SUMMIT MAX clinical trial, which will support regulatory submissions for the next-generation Monopoint Platform, a super-bore HiPoint 88 catheter delivered with Tenzing 8 for thrombectomy procedures.
Route 92 Medical intends to improve outcomes for patients undergoing neurovascular intervention through cutting-edge engineering and innovative product design. Founded by physicians, the company collaborates with neurovascular clinicians to solve the biggest challenges in neurointervention and deliver differentiated solutions.