Sam Brusco, Associate Editor06.20.23
Cardio Flow Design has earned U.S. Food and Drug Administration (FDA) approval for its iTFlow MRI blood flow analysis software to analyze blood flow using 4D Flow MRI data, potentially enhancing diagnostic accuracy for patient with cardiovascular diseases and heart conditions.
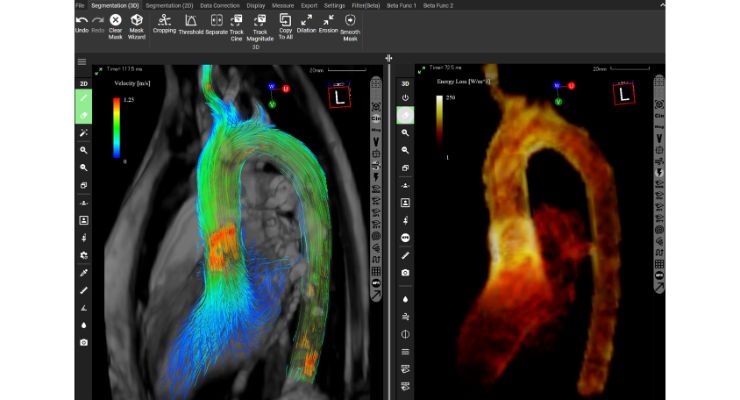
iTFlow software supports blood flow analysis in cardiovascular surgery and radiology. Using the tool, users can perform their own analysis and visualization of blood flow based on MRI data. Fluid dynamics in the heart and blood vessels can be visualized in both 2D and 3D, sometimes without using contrast agents.
Further, using iTFlow, physicians can make quantitative evaluations like flow rate, velocity, pathline, streamline, and other parameters.
Dr. Itatani, MD, Ph.D., a cardiovascular surgeon specializing in adult congenital heart disease and the inventor of iTFlow, told the press, "We developed this software with the aim of enabling all physicians, regardless of their level of experience, to propose the most appropriate treatment for patients even with complicated anatomy and cardiac function by quantitatively evaluating blood flow.”
Dr. Loke, associate professor of pediatrics at Children's National Hospital, who has been conducting retrospective blood flow analysis research with iTFlow before FDA approval, added: "Cardiac magnetic imaging research has demonstrated the emerging potential of advanced intracardiac flow analysis in heart disease beyond traditional measurements. To that end, iTFlow provides a front-line software interface to measure kinetic energies, vortexes, and shear stresses inside the heart without the need for complex in-house engineering software. I am excited by the potential of software such as iTFlow in bringing intracardiac flow analysis to clinical cardiology."
Cardio Flow Design CEO Teruyasu Nishino, MD, said, "We are delighted this software has obtained FDA approval. We have been pursuing the concept of blood flow analysis with 4D flow MRI for many years, and iTFlow encapsulates the essence of this concept. With this approval, we believe that healthcare professionals will be able to provide more accurate diagnoses and treatment plans for cardiovascular diseases, especially for children with congenital heart disease, and will save many lives. This innovation will change the way of diagnosis and serve as a catalyst to spread the adoption of blood flow analysis worldwide.”
iTFlow software supports blood flow analysis in cardiovascular surgery and radiology. Using the tool, users can perform their own analysis and visualization of blood flow based on MRI data. Fluid dynamics in the heart and blood vessels can be visualized in both 2D and 3D, sometimes without using contrast agents.
Further, using iTFlow, physicians can make quantitative evaluations like flow rate, velocity, pathline, streamline, and other parameters.
Dr. Itatani, MD, Ph.D., a cardiovascular surgeon specializing in adult congenital heart disease and the inventor of iTFlow, told the press, "We developed this software with the aim of enabling all physicians, regardless of their level of experience, to propose the most appropriate treatment for patients even with complicated anatomy and cardiac function by quantitatively evaluating blood flow.”
Dr. Loke, associate professor of pediatrics at Children's National Hospital, who has been conducting retrospective blood flow analysis research with iTFlow before FDA approval, added: "Cardiac magnetic imaging research has demonstrated the emerging potential of advanced intracardiac flow analysis in heart disease beyond traditional measurements. To that end, iTFlow provides a front-line software interface to measure kinetic energies, vortexes, and shear stresses inside the heart without the need for complex in-house engineering software. I am excited by the potential of software such as iTFlow in bringing intracardiac flow analysis to clinical cardiology."
Cardio Flow Design CEO Teruyasu Nishino, MD, said, "We are delighted this software has obtained FDA approval. We have been pursuing the concept of blood flow analysis with 4D flow MRI for many years, and iTFlow encapsulates the essence of this concept. With this approval, we believe that healthcare professionals will be able to provide more accurate diagnoses and treatment plans for cardiovascular diseases, especially for children with congenital heart disease, and will save many lives. This innovation will change the way of diagnosis and serve as a catalyst to spread the adoption of blood flow analysis worldwide.”