Sam Brusco, Associate Editor04.03.23
Masimo has gained De Novo approval from the U.S. Food and Drug Administration (FDA) for its Masimo Opioid Halo opioid overdose prevention and alert system.
The De Novo nod makes Opioid Halo the first and only FDA-cleared monitoring solution to detect opioid-induced respiratory depression, the leading cause of fatality from opioid overdose. Masimo also became the first winner of an FDA Opioid Innovation challenge to have an authorized product designed to help solve the U.S. opioid crisis.
Opioid Halo will be available over the counter (OTC) without a prescription, for use on adults and children 15 and older, as well as an Rx version for use with a prescription from a healthcare provider.
Its Opioid Halo engine is an advanced pattern recognition algorithm that helps detect and quantify risk of severe opioid-induced respiratory depression. Combined with its distributed architecture, it helps manage and send escalating alarms to family members, friends, and caregivers to notify them that help might be necessary due to an opioid overdose. This includes triggering an automatic wellness call, which might lead to EMS being dispatched.
Joe Kiani, founder and CEO of Masimo, told the press, “We are very excited to be able to offer this solution to our fellow Americans and the community heroes who are helping to battle the opioid crisis—a crisis so devastating in its impact on the young that it has lowered overall life expectancy in the U.S. Now, with Opioid Halo, we hope to help make a big difference by providing a much needed tool that can help millions, whether they are taking prescribed opioids or struggling with illicit opioid use. In 2018, we were honored to be chosen by the FDA, based on our expertise in patient monitoring technologies, to develop a device that could help prevent opioid overdose, as part of their Innovation Challenge addressing the opioid epidemic. And today, we are delighted to have received the first De Novo for our response to that Innovation Challenge, Masimo Opioid Halo. Between then and now, the COVID-19 pandemic accelerated our development and refinement of the Masimo SafetyNet family of remote patient management and telehealth solutions, helping save many COVID patients’ lives. We thank the FDA for taking on the opioid epidemic and granting this De Novo—a clearance that is a huge step forward in preventing overdose deaths and helping to end the opioid crisis.”
Opioid Halo is designed to help identify symptoms of an opioid overdose by spotting physiological markers present during opioid-induced respiratory depression and ideally, helping them know when it’s time to intervene—for example, by administering a potentially life-saving dose of naloxone. Opioid Halo can be used at home or in the hospital or another care setting, by patients prescribed opioids after surgery or managing a chronic or prolonged condition, as well as people suffering from opioid use disorder.
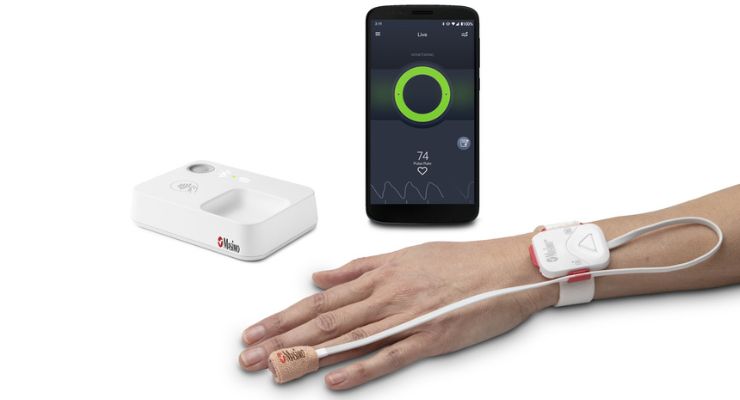
The system is comprised of a tetherless, adhesive fingertip sensor; a reusable Masimo SET pulse oximeter and Bluetooth chip; a Bluetooth-to-Wi-Fi Masimo Home Medical Hub; and a smartphone app.
The fingertip sensor offers real-time monitoring for opioid-induced respiratory depression, enabled by the pattern recognition algorithm and Masimo SET even during movement, when hands are cold, and on all skin pigmentations. Sensor and chip data are sent wirelessly to the Home Medical Hub and smartphone app, which continuously analyzes user physiological data for trends and patterns associated with opioid-induced respiratory depression.
As the risk level increases, the app and hub provide alerts. On early onset, an audible and visual alarm to trigger early intervention opportunities designed for the user to self-recover or get help, is provided. If the Opioid Halo risk score continues to worsen, automatic texts are sent to designated friends and family members. If the severity level progresses even further, there’s an optional setting that can be activated during setup that allows a service center to place a wellness call to the user, the outcome of which may lead to EMS being dispatched.
Kim Bennion, MsHS, RRT, CHC, FAARC, System Director of Research for Respiratory Care Clinical Services at Intermountain Health, a 33-hospital system based in Salt Lake City, Utah, said, “Painkillers often called ‘opioids’ can have negative side effects, including opioid-induced respiratory depression. Intermountain Health has demonstrated that home monitoring of post-surgical patients receiving opioids for pain can help identify issues earlier and avoid adverse outcomes. As a registered respiratory therapist and leader in the field of opioid-induced respiratory depression in the post-surgical setting, we are excited that Masimo’s breakthrough device will be available for our patients.”
The De Novo nod makes Opioid Halo the first and only FDA-cleared monitoring solution to detect opioid-induced respiratory depression, the leading cause of fatality from opioid overdose. Masimo also became the first winner of an FDA Opioid Innovation challenge to have an authorized product designed to help solve the U.S. opioid crisis.
Opioid Halo will be available over the counter (OTC) without a prescription, for use on adults and children 15 and older, as well as an Rx version for use with a prescription from a healthcare provider.
Its Opioid Halo engine is an advanced pattern recognition algorithm that helps detect and quantify risk of severe opioid-induced respiratory depression. Combined with its distributed architecture, it helps manage and send escalating alarms to family members, friends, and caregivers to notify them that help might be necessary due to an opioid overdose. This includes triggering an automatic wellness call, which might lead to EMS being dispatched.
Joe Kiani, founder and CEO of Masimo, told the press, “We are very excited to be able to offer this solution to our fellow Americans and the community heroes who are helping to battle the opioid crisis—a crisis so devastating in its impact on the young that it has lowered overall life expectancy in the U.S. Now, with Opioid Halo, we hope to help make a big difference by providing a much needed tool that can help millions, whether they are taking prescribed opioids or struggling with illicit opioid use. In 2018, we were honored to be chosen by the FDA, based on our expertise in patient monitoring technologies, to develop a device that could help prevent opioid overdose, as part of their Innovation Challenge addressing the opioid epidemic. And today, we are delighted to have received the first De Novo for our response to that Innovation Challenge, Masimo Opioid Halo. Between then and now, the COVID-19 pandemic accelerated our development and refinement of the Masimo SafetyNet family of remote patient management and telehealth solutions, helping save many COVID patients’ lives. We thank the FDA for taking on the opioid epidemic and granting this De Novo—a clearance that is a huge step forward in preventing overdose deaths and helping to end the opioid crisis.”
Opioid Halo is designed to help identify symptoms of an opioid overdose by spotting physiological markers present during opioid-induced respiratory depression and ideally, helping them know when it’s time to intervene—for example, by administering a potentially life-saving dose of naloxone. Opioid Halo can be used at home or in the hospital or another care setting, by patients prescribed opioids after surgery or managing a chronic or prolonged condition, as well as people suffering from opioid use disorder.
The system is comprised of a tetherless, adhesive fingertip sensor; a reusable Masimo SET pulse oximeter and Bluetooth chip; a Bluetooth-to-Wi-Fi Masimo Home Medical Hub; and a smartphone app.
The fingertip sensor offers real-time monitoring for opioid-induced respiratory depression, enabled by the pattern recognition algorithm and Masimo SET even during movement, when hands are cold, and on all skin pigmentations. Sensor and chip data are sent wirelessly to the Home Medical Hub and smartphone app, which continuously analyzes user physiological data for trends and patterns associated with opioid-induced respiratory depression.
As the risk level increases, the app and hub provide alerts. On early onset, an audible and visual alarm to trigger early intervention opportunities designed for the user to self-recover or get help, is provided. If the Opioid Halo risk score continues to worsen, automatic texts are sent to designated friends and family members. If the severity level progresses even further, there’s an optional setting that can be activated during setup that allows a service center to place a wellness call to the user, the outcome of which may lead to EMS being dispatched.
Kim Bennion, MsHS, RRT, CHC, FAARC, System Director of Research for Respiratory Care Clinical Services at Intermountain Health, a 33-hospital system based in Salt Lake City, Utah, said, “Painkillers often called ‘opioids’ can have negative side effects, including opioid-induced respiratory depression. Intermountain Health has demonstrated that home monitoring of post-surgical patients receiving opioids for pain can help identify issues earlier and avoid adverse outcomes. As a registered respiratory therapist and leader in the field of opioid-induced respiratory depression in the post-surgical setting, we are excited that Masimo’s breakthrough device will be available for our patients.”