Michael Barbella, Managing Editor03.29.23
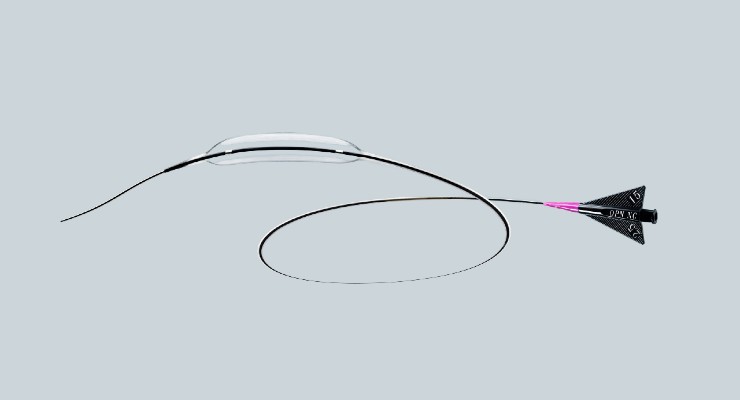
SIS Medical AG (Swiss Interventional Systems) has launched its OPN NC PTCA Dilatation Catheter with TWIN-Wall technology in the United States. The product extends treatment options by high performance and safety as well as providing success rates where other balloons fail.
The catheter is being distributed by Lenexa, Kan.-based Worldwide Innovations & Technologies Inc. (manufacturer and distributor of RADPAD Radiation Protection Products).
The OPN NC catheter features a TWIN-Wall design, which provides a super-high pressure resistance with a rated burst pressure of 35 atm with a very low compliance. These performance characteristics allow for a perfect preparation of challenging lesions prior to stenting without over-dilating of the blood vessel. The device can also be applied for post-dilatation of under-expanded stents. OPN NC coronary balloon is the only Super-High-Pressure coronary balloon rated with 35 atm which received U.S. Food and Drug Administration 510(k) clearance (March 2022).
The first U.S. treatment with the catheter occurred at Morristown Medical Center (New Jersey) by interventional cardiologist Dr. Philippe Genereux. “I was extremely satisfied with the performance of the OPN NC balloon. We were successful in efficiently treating multiple patients with very complex and calcified lesions," he said. "The OPN NC balloon is an essential tool for operators treating resistant lesions, restenosis, or to treat acutely under-expanded stent, and is a true cost-effective technology compared to other devices.”
“Our OPN NC balloon with TWIN-Wall technology has already been successfully used for several years in Europe and other countries. More than 140,000 patients have been treated with the product so far. With the U.S. launch, our company has reached yet another significant milestone. The first cases at the Morristown Medical Center have shown a strong need for our technology to improve the treatment of challenging coronary lesions in US patients. Many other US hospitals will be introducing the OPN NC,” SIS Medical AG CEO Sergej Kammerzell stated.
“Access to the U.S. market will have a great impact on the further development, reputation, and continued international growth of SIS Medical AG,” adds Willi Zwahlen, vice president of Sales & Marketing, founder, and SIS Medical board member. “The U.S. launch is a key-element of our strategy to make OPN NC available in all major global markets.”
“OPN NC is an exciting product that we can provide to a wide customer base in the U.S. We look forward to adding value to our customer facilities by delivering another life-saving device,” Eric Radtke, vice president of Sales & Marketing at RADPAD said.
“We are excited to be the U.S. distributor for OPN NC and look forward to being a part of providing a major step forward in interventional medicine in the U.S.,” RADPAD CEO John Cadwalader stated.
SIS Medical AG, based in Frauenfeld, Switzerland, develops, manufactures, and distributes high-performance PTCA balloons.
Worldwide Innovations & Technologies Inc., founded in July 1992, develops and markets radiation protection products used in coronary catheter labs. RADPAD emerged from WIT’s developmental years.
The catheter is being distributed by Lenexa, Kan.-based Worldwide Innovations & Technologies Inc. (manufacturer and distributor of RADPAD Radiation Protection Products).
The OPN NC catheter features a TWIN-Wall design, which provides a super-high pressure resistance with a rated burst pressure of 35 atm with a very low compliance. These performance characteristics allow for a perfect preparation of challenging lesions prior to stenting without over-dilating of the blood vessel. The device can also be applied for post-dilatation of under-expanded stents. OPN NC coronary balloon is the only Super-High-Pressure coronary balloon rated with 35 atm which received U.S. Food and Drug Administration 510(k) clearance (March 2022).
The first U.S. treatment with the catheter occurred at Morristown Medical Center (New Jersey) by interventional cardiologist Dr. Philippe Genereux. “I was extremely satisfied with the performance of the OPN NC balloon. We were successful in efficiently treating multiple patients with very complex and calcified lesions," he said. "The OPN NC balloon is an essential tool for operators treating resistant lesions, restenosis, or to treat acutely under-expanded stent, and is a true cost-effective technology compared to other devices.”
“Our OPN NC balloon with TWIN-Wall technology has already been successfully used for several years in Europe and other countries. More than 140,000 patients have been treated with the product so far. With the U.S. launch, our company has reached yet another significant milestone. The first cases at the Morristown Medical Center have shown a strong need for our technology to improve the treatment of challenging coronary lesions in US patients. Many other US hospitals will be introducing the OPN NC,” SIS Medical AG CEO Sergej Kammerzell stated.
“Access to the U.S. market will have a great impact on the further development, reputation, and continued international growth of SIS Medical AG,” adds Willi Zwahlen, vice president of Sales & Marketing, founder, and SIS Medical board member. “The U.S. launch is a key-element of our strategy to make OPN NC available in all major global markets.”
“OPN NC is an exciting product that we can provide to a wide customer base in the U.S. We look forward to adding value to our customer facilities by delivering another life-saving device,” Eric Radtke, vice president of Sales & Marketing at RADPAD said.
“We are excited to be the U.S. distributor for OPN NC and look forward to being a part of providing a major step forward in interventional medicine in the U.S.,” RADPAD CEO John Cadwalader stated.
SIS Medical AG, based in Frauenfeld, Switzerland, develops, manufactures, and distributes high-performance PTCA balloons.
Worldwide Innovations & Technologies Inc., founded in July 1992, develops and markets radiation protection products used in coronary catheter labs. RADPAD emerged from WIT’s developmental years.