Sam Brusco, Associate Editor02.14.23
HistoSonics, developer of the histotripsy non-invasive platform and sonic beam therapy, has been granted U.S. Food and Drug Administration (FDA) approval for the company-sponsored #HOPE4KIDNEY trial.
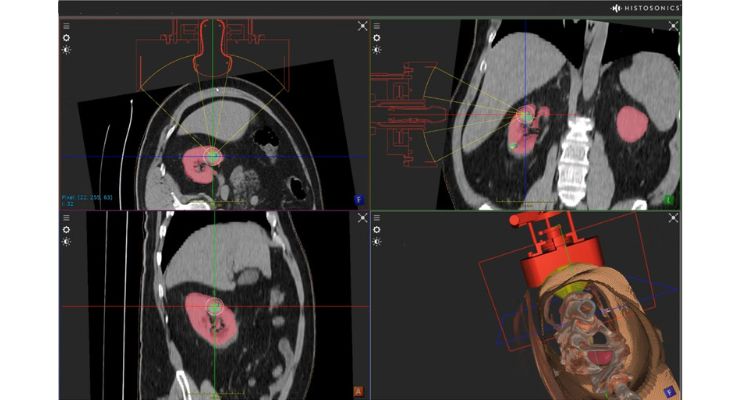
The trial aims to assess safety and technical success of the company’s Edison system to target and destroy targeted primary renal tumors non-invasively and without incisions or needles. The FDA approval for the trial follows HistoSonics’ recent submission to the agency from its #HOPE4LIVER trials, where both primary endpoints of safety and efficacy were met in destroying liver tumors.
"We are very pleased with the FDA's approval of our #HOPE4KIDNEY Trial and appreciative of the agency's prompt review process. This approval represents a substantial milestone for our company as we continue to expand histotripsy and its potential benefits into diseases that impact the lives of so many people," Mike Blue, president and CEO of HistoSonics told the press.
He added, “We are excited to expand on our experiences in successfully targeting and treating in the liver using our enhanced Edison platform that combines advanced imaging and targeting capabilities with real time treatment monitoring. The kidney is a logical next application as treating in the kidney has very similar procedural and anatomical considerations as the liver, and Edison was specifically designed to treat anywhere in the abdomen, as a starting point. In addition, the prevalence of kidney disease remains high with many patients kept in active surveillance or watchful waiting."
Surgical intervention is currently the gold standard to remove kidney tumors but a non-invasive approach using histotripsy can destroy targeted tissue without damaging non-targeted kidney tissue. Further, the tech’s mechanism of cellular destruction may preserve function of the kidney’s urine collecting system and remove some complications seen in existing invasive procedures.
The company’s image-guided sonic beam therapy uses advanced imaging and proprietary sensing tech for non-invasive personalized treatment. Histotripsy uses focused sound energy to create controlled acoustic cavitation that mechanically destroys and liquefies targeted liver tissue at sub-cellular levels.
HistoSonics believes the novel mechanism of action allows the treatment site to recover and resorb quickly. Physicians can also monitor destruction of liver tissue under continuous real-time visualization and control.
The Edison system is currently pending FDA review for a destruction of liver tissue indication. The #HOPE4KIDNEY trial aims to support future indication expansion to include destruction of kidney tissue.
The trial aims to assess safety and technical success of the company’s Edison system to target and destroy targeted primary renal tumors non-invasively and without incisions or needles. The FDA approval for the trial follows HistoSonics’ recent submission to the agency from its #HOPE4LIVER trials, where both primary endpoints of safety and efficacy were met in destroying liver tumors.
"We are very pleased with the FDA's approval of our #HOPE4KIDNEY Trial and appreciative of the agency's prompt review process. This approval represents a substantial milestone for our company as we continue to expand histotripsy and its potential benefits into diseases that impact the lives of so many people," Mike Blue, president and CEO of HistoSonics told the press.
He added, “We are excited to expand on our experiences in successfully targeting and treating in the liver using our enhanced Edison platform that combines advanced imaging and targeting capabilities with real time treatment monitoring. The kidney is a logical next application as treating in the kidney has very similar procedural and anatomical considerations as the liver, and Edison was specifically designed to treat anywhere in the abdomen, as a starting point. In addition, the prevalence of kidney disease remains high with many patients kept in active surveillance or watchful waiting."
Surgical intervention is currently the gold standard to remove kidney tumors but a non-invasive approach using histotripsy can destroy targeted tissue without damaging non-targeted kidney tissue. Further, the tech’s mechanism of cellular destruction may preserve function of the kidney’s urine collecting system and remove some complications seen in existing invasive procedures.
The company’s image-guided sonic beam therapy uses advanced imaging and proprietary sensing tech for non-invasive personalized treatment. Histotripsy uses focused sound energy to create controlled acoustic cavitation that mechanically destroys and liquefies targeted liver tissue at sub-cellular levels.
HistoSonics believes the novel mechanism of action allows the treatment site to recover and resorb quickly. Physicians can also monitor destruction of liver tissue under continuous real-time visualization and control.
The Edison system is currently pending FDA review for a destruction of liver tissue indication. The #HOPE4KIDNEY trial aims to support future indication expansion to include destruction of kidney tissue.