Sam Brusco, Associate Editor11.29.22
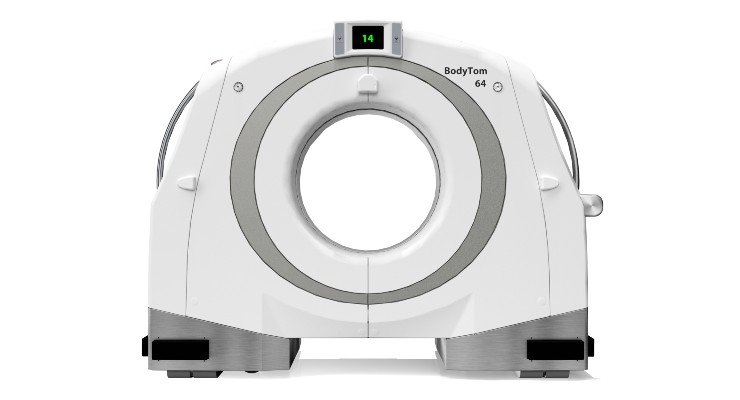
Samsung subsidiary NeuroLogica has earned U.S. Food and Drug Administration (FDA) 510(k) clearance for its BodyTom 64 point-of-care mobile CT scanner, a head-to-toe trauma imaging solution.
“We’re thrilled to build off our expertise and elevate point-of-care imaging with our BodyTom 64, which can transform any room in a hospital into an advanced imaging suite,” Jason Koshnitsky, sr. director of global sales and marketing of NeuroLogica told the press. “This full-body 64-slice CT scanner is an upgraded version of the BodyTom Elite CT scanner, providing enhanced functionality with the same high-resolution imaging capabilities.”
Revisions to the software and hardware include Linux as the operating system and the ability to generate up to 64 cross-sectional CT images of a patient’s body.
The multi-departmental imaging solution can be used for neurosurgery/surgery, trauma/ER, and interventional radiology.
“We’re thrilled to build off our expertise and elevate point-of-care imaging with our BodyTom 64, which can transform any room in a hospital into an advanced imaging suite,” Jason Koshnitsky, sr. director of global sales and marketing of NeuroLogica told the press. “This full-body 64-slice CT scanner is an upgraded version of the BodyTom Elite CT scanner, providing enhanced functionality with the same high-resolution imaging capabilities.”
Revisions to the software and hardware include Linux as the operating system and the ability to generate up to 64 cross-sectional CT images of a patient’s body.
The multi-departmental imaging solution can be used for neurosurgery/surgery, trauma/ER, and interventional radiology.