Sam Brusco, Associate Editor10.11.22
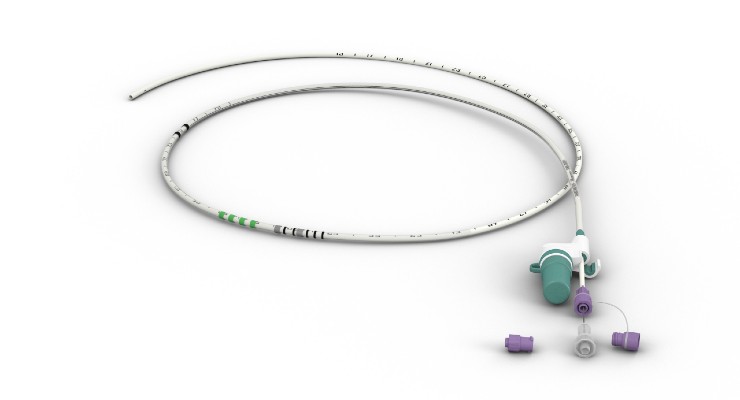
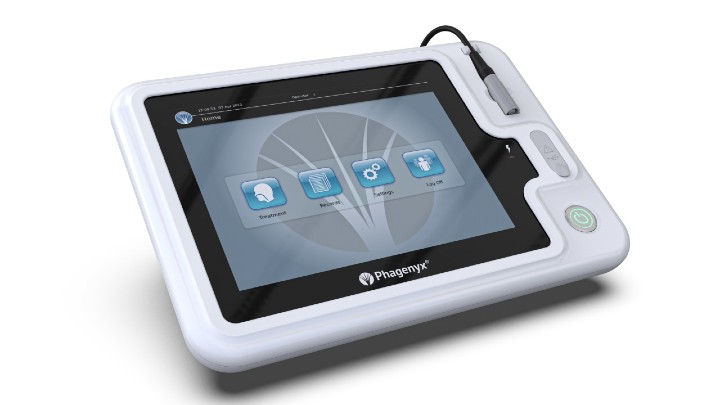
Phagenesis has received de novo approval from the U.S. Food and Drug Administration (FDA) for its Phagenyx neurostimulation system, a therapy that uses pharyngeal electrical stimulation to restore swallowing control in those with severe dysphagia post-stroke. The company is preparing a targeted U.S. launch.
The most common reason for neurogenic dysphagia is central nervous system injury due to stroke or traumatic brain injury. It can also be caused by peripheral nervous system damage due to prolonged mechanical ventilation or tracheostomy.
"Dysphagia affects millions of people worldwide and many do not receive the desired outcome with currently available treatments," Marta Kazandjian, MA CCC-SLP BCS-S, F-ASHA and Department Head for Speech Language Pathology and Swallowing Services at Stony Brook Southampton Hospital, told the press. "Adding an innovative therapy like Phagenyx has the potential to accelerate results and may allow the patient to spend fewer days in the hospital, which would significantly improve quality of care and patient experience."
Phagenyx’s pharyngeal electrical stimulation (PES) targets the neurological components of swallowing coordination. Phagenyx therapy has shown best results when delivered at the earliest stages after injury to enhance neurorehabilitation and restore swallowing control.
"There is limited awareness of the significance of dysphagia in the critical care setting following prolonged mechanical ventilation or tracheostomy," said Dr. Wade Smith, Professor of Neurology at the University of California, San Francisco (UCSF). "Such patients cannot manage their own saliva, creating a high risk for aspiration and their safety. Phagenyx has the potential to treat patients early in the ICU which has been shown to accelerate recovery, reduce hospital length of stay and prevent downstream complications."
"The Phagenyx System is a next-generation therapy for neurogenic dysphagia that has been shown to deliver significant benefits in swallowing safety and swallowing efficiency, which have the potential to translate into substantial health economic benefits due to accelerated patient recovery," commented Reinhard Krickl, CEO of Phagenesis. "We are pleased with FDA's decision to grant our de novo request and are eager to bring this new technology to the millions of patients in the U.S. who suffer from post-stroke dysphagia and are desperate for a better solution."
The most common reason for neurogenic dysphagia is central nervous system injury due to stroke or traumatic brain injury. It can also be caused by peripheral nervous system damage due to prolonged mechanical ventilation or tracheostomy.
"Dysphagia affects millions of people worldwide and many do not receive the desired outcome with currently available treatments," Marta Kazandjian, MA CCC-SLP BCS-S, F-ASHA and Department Head for Speech Language Pathology and Swallowing Services at Stony Brook Southampton Hospital, told the press. "Adding an innovative therapy like Phagenyx has the potential to accelerate results and may allow the patient to spend fewer days in the hospital, which would significantly improve quality of care and patient experience."
Phagenyx’s pharyngeal electrical stimulation (PES) targets the neurological components of swallowing coordination. Phagenyx therapy has shown best results when delivered at the earliest stages after injury to enhance neurorehabilitation and restore swallowing control.

"There is limited awareness of the significance of dysphagia in the critical care setting following prolonged mechanical ventilation or tracheostomy," said Dr. Wade Smith, Professor of Neurology at the University of California, San Francisco (UCSF). "Such patients cannot manage their own saliva, creating a high risk for aspiration and their safety. Phagenyx has the potential to treat patients early in the ICU which has been shown to accelerate recovery, reduce hospital length of stay and prevent downstream complications."
"The Phagenyx System is a next-generation therapy for neurogenic dysphagia that has been shown to deliver significant benefits in swallowing safety and swallowing efficiency, which have the potential to translate into substantial health economic benefits due to accelerated patient recovery," commented Reinhard Krickl, CEO of Phagenesis. "We are pleased with FDA's decision to grant our de novo request and are eager to bring this new technology to the millions of patients in the U.S. who suffer from post-stroke dysphagia and are desperate for a better solution."