Samuel Brusco, Associate Editor09.16.22
Abiomed has received two U.S. Food and Drug Administration (FDA) approvals for clinical research of its Impella heart pumps for acute myocardial infarction (AMI) cardiogenic shock.
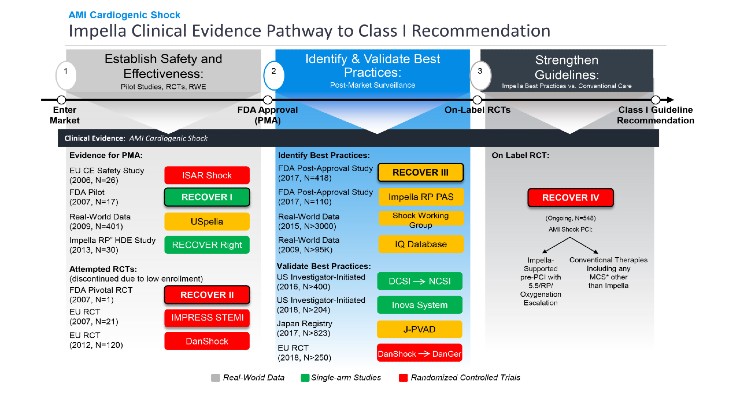
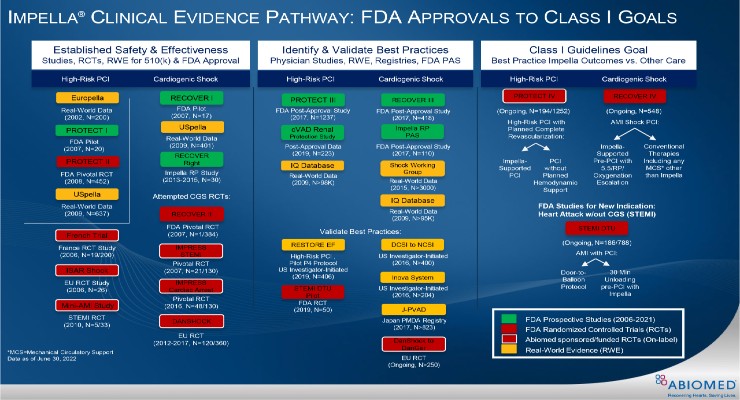
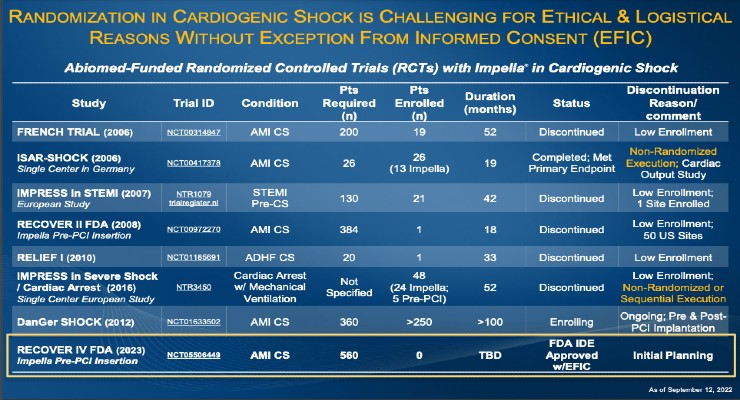
The first approval is for the on-label RECOVER IV trial for AMI cardiogenic shock. The two-arm trial will evaluate if percutaneous coronary intervention (PCI) with Impella support prior to the PCI is superior to PCI without Impella.
“This landmark trial will be the culmination of over 20 years of research in the interventional therapy of AMI and will apply all the clinical advancements we have made to improve survival and heart recovery for AMI patients with cardiogenic shock as demonstrated in multiple prospective studies,” William O’Neill, MD, medical director of the Center for Structural Heart Disease at Henry Ford Health and a RECOVER IV national co-principal investigator, told the press.
The trial’s primary endpoint is all-cause mortality at 30 days. Secondary endpoints are major adverse cardiovascular and cerebrovascular events (MACCE) at 30 days, days alive out of the hospital at six months, recovery of left ventricular (LV) function, need for durable ventricular assist device (VAD) or heart transplant, and health-related quality of life as measured by responses to the Kansas City Cardiomyopathy Questionnaire (KCCQ) at one year.
Abiomed aims to achieve global AMI cardiogenic shock Class I guideline recommendation for Impella.
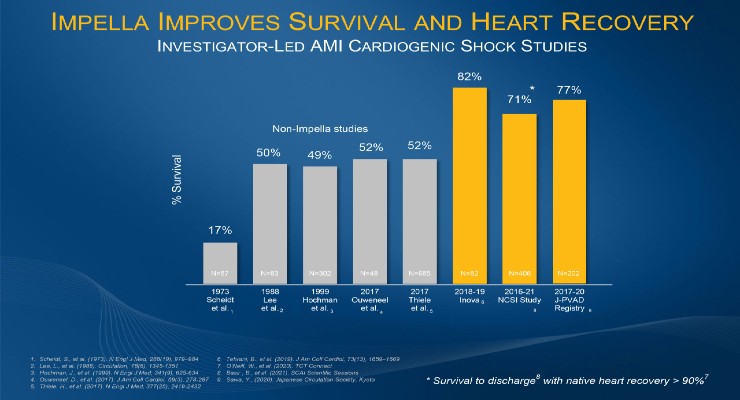
“I am optimistic that RECOVER IV will further demonstrate the benefits of hemodynamic support and best practice protocols. These benefits include ventricular unloading using Impella pre-PCI, reduced LV wall stress, reduced pulmonary congestion, increased collateral coronary blood flow, and enhanced cardio protection so that more AMI cardiogenic shock patients can survive and achieve native heart recovery. The heart team and field have evolved and understand how important myocardial recovery is for both AMI and AMI cardiogenic shock to reduce the growing epidemic of heart failure,” said Navin K. Kapur, MD, executive director of the Cardiovascular Center for Research and Innovation (CVCRI) at Tufts Medical Center and a national co-principal investigator for RECOVER IV.
The FDA also approved and closed Impella’s RECOVER III prospective AMI cardiogenic shock post-approval study. The study-gathered real-world evidence on AMI cardiogenic shock treated with Impella between 2107-2019.
According to the company, Impella remains the only mechanical circulatory support (MCS) device that has received the FDA’s highest level of premarket approval (PMA) and PAS regulatory approval for AMI cardiogenic shock. Based on RECOVER III, Impella’s label for AMI cardiogenic shock will be updated to reflect data for up to one-year post-procedure.
The first approval is for the on-label RECOVER IV trial for AMI cardiogenic shock. The two-arm trial will evaluate if percutaneous coronary intervention (PCI) with Impella support prior to the PCI is superior to PCI without Impella.
“This landmark trial will be the culmination of over 20 years of research in the interventional therapy of AMI and will apply all the clinical advancements we have made to improve survival and heart recovery for AMI patients with cardiogenic shock as demonstrated in multiple prospective studies,” William O’Neill, MD, medical director of the Center for Structural Heart Disease at Henry Ford Health and a RECOVER IV national co-principal investigator, told the press.
The trial’s primary endpoint is all-cause mortality at 30 days. Secondary endpoints are major adverse cardiovascular and cerebrovascular events (MACCE) at 30 days, days alive out of the hospital at six months, recovery of left ventricular (LV) function, need for durable ventricular assist device (VAD) or heart transplant, and health-related quality of life as measured by responses to the Kansas City Cardiomyopathy Questionnaire (KCCQ) at one year.
Abiomed aims to achieve global AMI cardiogenic shock Class I guideline recommendation for Impella.
“I am optimistic that RECOVER IV will further demonstrate the benefits of hemodynamic support and best practice protocols. These benefits include ventricular unloading using Impella pre-PCI, reduced LV wall stress, reduced pulmonary congestion, increased collateral coronary blood flow, and enhanced cardio protection so that more AMI cardiogenic shock patients can survive and achieve native heart recovery. The heart team and field have evolved and understand how important myocardial recovery is for both AMI and AMI cardiogenic shock to reduce the growing epidemic of heart failure,” said Navin K. Kapur, MD, executive director of the Cardiovascular Center for Research and Innovation (CVCRI) at Tufts Medical Center and a national co-principal investigator for RECOVER IV.
The FDA also approved and closed Impella’s RECOVER III prospective AMI cardiogenic shock post-approval study. The study-gathered real-world evidence on AMI cardiogenic shock treated with Impella between 2107-2019.
According to the company, Impella remains the only mechanical circulatory support (MCS) device that has received the FDA’s highest level of premarket approval (PMA) and PAS regulatory approval for AMI cardiogenic shock. Based on RECOVER III, Impella’s label for AMI cardiogenic shock will be updated to reflect data for up to one-year post-procedure.