Michael Barbella, Managing Editor06.02.22
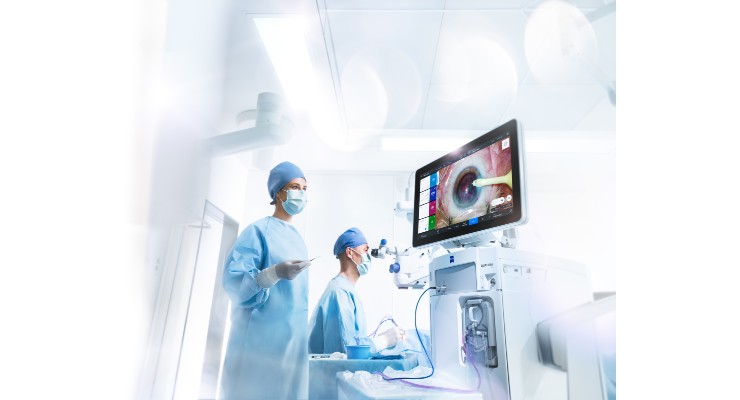
ZEISS Medical Technology has received U.S. Food and Drug Administration (FDA) clearance for the QUATERA 700,* an advancement in phaco technology. The QUATERA 700 includes the QUATTRO Pump, which delivers exceptional chamber stability independent of intra-ocular pressure (IOP) and flow.
“What excites me the most about the QUATERA 700 is the QUATTRO pump’s synchronized fluidics system and how I can have both the highest levels of efficiency, while maintaining an extremely stable chamber, independent of IOP,” said Sri Ganesh, M.D., chairman and managing director of Nethradhama Super Speciality Eye Hospital in Bangalore (India).
The ZEISS QUATERA 700 is designed to increase a surgeon’s workflow efficiency from the clinic to the operating room (OR), enabling one digitally integrated surgical workflow. It works as a single sterile OR dashboard integrating patient data from other systems and the microscope view, available in real-time for the OR staff.
“It’s great to see that ZEISS has added another critical piece to the cataract workflow, a digital solution that further enables us to become more efficient and effective during surgical procedures,” said Elizabeth Yeu, M.D., of Virginia Eye Consultants in Norfolk.
“Building on the company’s medical technologies, ZEISS is committed to continuously expanding our solutions,” said Dr. Markus Weber, president and CEO of Carl Zeiss Meditec AG. “Our investment in the latest digital technologies help make customers’ workflows more efficient by integrating solutions and improving clinical results.”
“ZEISS is committed to bringing our latest innovations to the United States, and the ZEISS QUATERA 700 is a great example of how integrated technology can drive efficiency in ophthalmology,” said Euan S. Thomson, Ph.D., president of Ophthalmic Devices and Head of the Digital Business Unit for Carl Zeiss Meditec. “Together with our ZEISS Medical Ecosystem and surgical planning software, we believe this fully connected and integrated approach will help set new standards for patient care and clinical management.”
Carl Zeiss Meditec AG is a global medical technology company that supplies technologies and application-oriented solutions, including implants and consumables, to diagnose and treat eye diseases. The company creates visualization solutions in the field of microsurgery. With approximately 3,531 employees worldwide, the Group generated 1.64 billion euros in fiscal year 2020/21. The Group’s head office is located in Jena, Germany, and it has subsidiaries in Germany and abroad; more than 50 percent of its employees are based in the United States, Japan, Spain, and France. The Center for Application and Research (CARIn) in Bangalore, India, and the Carl Zeiss Innovations Center for Research and Development in Shanghai, China, strengthen the company's presence in these rapidly developing economies. Around 41 percent of Carl Zeiss Meditec AG’s shares are in free float. The remaining approx. 59 percent are held by Carl Zeiss AG.
“What excites me the most about the QUATERA 700 is the QUATTRO pump’s synchronized fluidics system and how I can have both the highest levels of efficiency, while maintaining an extremely stable chamber, independent of IOP,” said Sri Ganesh, M.D., chairman and managing director of Nethradhama Super Speciality Eye Hospital in Bangalore (India).
The ZEISS QUATERA 700 is designed to increase a surgeon’s workflow efficiency from the clinic to the operating room (OR), enabling one digitally integrated surgical workflow. It works as a single sterile OR dashboard integrating patient data from other systems and the microscope view, available in real-time for the OR staff.
“It’s great to see that ZEISS has added another critical piece to the cataract workflow, a digital solution that further enables us to become more efficient and effective during surgical procedures,” said Elizabeth Yeu, M.D., of Virginia Eye Consultants in Norfolk.
“Building on the company’s medical technologies, ZEISS is committed to continuously expanding our solutions,” said Dr. Markus Weber, president and CEO of Carl Zeiss Meditec AG. “Our investment in the latest digital technologies help make customers’ workflows more efficient by integrating solutions and improving clinical results.”
“ZEISS is committed to bringing our latest innovations to the United States, and the ZEISS QUATERA 700 is a great example of how integrated technology can drive efficiency in ophthalmology,” said Euan S. Thomson, Ph.D., president of Ophthalmic Devices and Head of the Digital Business Unit for Carl Zeiss Meditec. “Together with our ZEISS Medical Ecosystem and surgical planning software, we believe this fully connected and integrated approach will help set new standards for patient care and clinical management.”
Carl Zeiss Meditec AG is a global medical technology company that supplies technologies and application-oriented solutions, including implants and consumables, to diagnose and treat eye diseases. The company creates visualization solutions in the field of microsurgery. With approximately 3,531 employees worldwide, the Group generated 1.64 billion euros in fiscal year 2020/21. The Group’s head office is located in Jena, Germany, and it has subsidiaries in Germany and abroad; more than 50 percent of its employees are based in the United States, Japan, Spain, and France. The Center for Application and Research (CARIn) in Bangalore, India, and the Carl Zeiss Innovations Center for Research and Development in Shanghai, China, strengthen the company's presence in these rapidly developing economies. Around 41 percent of Carl Zeiss Meditec AG’s shares are in free float. The remaining approx. 59 percent are held by Carl Zeiss AG.